If you received monoclonal antibody (mAb) therapy or convalescent plasma after exposure or infection, you do NOT need to delay your COVID-19 vaccination. Monoclonal antibodies are supplemental antibodies that can be administered early in the course of infection the first 10 days after symptoms To receive email updates about COVID-19, enter your email address: We take your privacy seriously. Where can I find a drive-thru vaccine site? Please note that any UCHealth Urgent Care location can evaluate you in order to determine if you qualify. University of Alabama at BirminghamProfessor Turner Overton, M.D., says the infusion is effective; but people still need to be vaccinated to receive the strongest protection against hospitalization due to COVID-19.  In November 2022, the CDC reported on a real-world study that showed adults who took Paxlovid within five days of a COVID-19 diagnosis had a 51% lower hospitalization rate within the next 30 days than those who were not given the drug. [Originally published: March 10, 2022. Ginde said it can be a life-saving treatment when administered in time. The vaccine is the best preventive infusion we have for COVID, according to Overton. Necessary cookies are absolutely essential for the website to function properly. UCHealth is encouraging people at risk of getting very sick from COVID-19 to test as soon as they detect symptoms. A CDC study reported that rebound symptoms tended to be unlikely to require hospitalization and milder than symptoms experienced during the primary infection. Mine was 8 days after first symptoms, but I didn't get a positive test til a day before treatment. The monoclonal antibody treatments are meant for mild to moderate COVID cases in adults and children over 12 to prevent the progression of severe COVID. Please get vaccinated, Overton said. 221 0 obj
<>/Filter/FlateDecode/ID[<9E92E7771DBCAB47966B3E43AFE6176F>]/Index[203 36]/Info 202 0 R/Length 86/Prev 80370/Root 204 0 R/Size 239/Type/XRef/W[1 2 1]>>stream
Youll hear not infrequently reports of people that are that sick that within even six to 12 hours feeling like theyve taken a dramatic turn to the better., The earlier, the better, Ginde said. New findings from a study of thousands of healthcare workers in England show that those who got COVID-19 and produced antibodies against the virus are highly unlikely to become infected again, at least over the 84-86 Similarly, protective antibody titres are maintained in patients undergoing anti-CD19 CAR T-cell therapy for B-cell malignancy. Because a monoclonal antibody treatment may interfere with a vaccine-induced immune response, the CDC recommends waiting at least 90 days before getting a COVID vaccine after you receive treatment. These cookies allow us to count visits and traffic sources so we can measure and improve the performance of our site.
In November 2022, the CDC reported on a real-world study that showed adults who took Paxlovid within five days of a COVID-19 diagnosis had a 51% lower hospitalization rate within the next 30 days than those who were not given the drug. [Originally published: March 10, 2022. Ginde said it can be a life-saving treatment when administered in time. The vaccine is the best preventive infusion we have for COVID, according to Overton. Necessary cookies are absolutely essential for the website to function properly. UCHealth is encouraging people at risk of getting very sick from COVID-19 to test as soon as they detect symptoms. A CDC study reported that rebound symptoms tended to be unlikely to require hospitalization and milder than symptoms experienced during the primary infection. Mine was 8 days after first symptoms, but I didn't get a positive test til a day before treatment. The monoclonal antibody treatments are meant for mild to moderate COVID cases in adults and children over 12 to prevent the progression of severe COVID. Please get vaccinated, Overton said. 221 0 obj
<>/Filter/FlateDecode/ID[<9E92E7771DBCAB47966B3E43AFE6176F>]/Index[203 36]/Info 202 0 R/Length 86/Prev 80370/Root 204 0 R/Size 239/Type/XRef/W[1 2 1]>>stream
Youll hear not infrequently reports of people that are that sick that within even six to 12 hours feeling like theyve taken a dramatic turn to the better., The earlier, the better, Ginde said. New findings from a study of thousands of healthcare workers in England show that those who got COVID-19 and produced antibodies against the virus are highly unlikely to become infected again, at least over the 84-86 Similarly, protective antibody titres are maintained in patients undergoing anti-CD19 CAR T-cell therapy for B-cell malignancy. Because a monoclonal antibody treatment may interfere with a vaccine-induced immune response, the CDC recommends waiting at least 90 days before getting a COVID vaccine after you receive treatment. These cookies allow us to count visits and traffic sources so we can measure and improve the performance of our site. 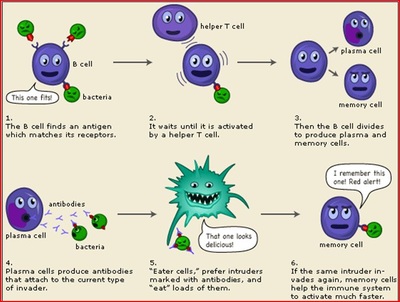 Another big difference is that while there is a small window of time to get this COVID treatment, the COVID vaccines will always have the memory cells to produce the antibodies immediately. This rate reflects updated information about the costs involved in furnishing these complex products in a patients home. Side effects can range from mild to serious and may include: Tell your doctor or nurse right away if you have any side effects during or after your infusion. For patients with severe kidney diseaseor who are on dialysisor those with severe liver disease, Paxlovid is not recommended; the levels of the drug can become too high and could cause increased side effects, he says. UAB also encourages applications from individuals with disabilities and veterans. (The FDA has provided a fact sheet on Paxlovid with a full list of known side effects.). "If you received monoclonal antibody treatment, such as Regeneron or sotrovimab, you need to wait 90 days after infusion Comparing the COVID-19 Vaccines: How Are They Different? Both are prescription-only oral antiviral pills given early in illness. %PDF-1.5
%
Its always been the Achilles heel of these antiviral drugs that most people dont get testedor they dont have access to testing.. Sanitiza tu hogar o negocio con los mejores resultados. Performance cookies are used to understand and analyze the key performance indexes of the website which helps in delivering a better user experience for the visitors. By entering your email and clicking Sign Up, you're agreeing to let us send you customized marketing messages about us and our advertising partners. Most people that test positive for symptomatic COVID-19 are actually eligible for this treatment because they have one or more risk factors for severe disease, but the vast majority of them do not even know about this treatment, said Adit Ginde, an epidemiologist at the University of Colorado School of Medicine and an emergency department physician at UCHealth, a Colorado-based health system. But dont expect to have the protection of monoclonal antibodies for those full 90 days in your body.
Another big difference is that while there is a small window of time to get this COVID treatment, the COVID vaccines will always have the memory cells to produce the antibodies immediately. This rate reflects updated information about the costs involved in furnishing these complex products in a patients home. Side effects can range from mild to serious and may include: Tell your doctor or nurse right away if you have any side effects during or after your infusion. For patients with severe kidney diseaseor who are on dialysisor those with severe liver disease, Paxlovid is not recommended; the levels of the drug can become too high and could cause increased side effects, he says. UAB also encourages applications from individuals with disabilities and veterans. (The FDA has provided a fact sheet on Paxlovid with a full list of known side effects.). "If you received monoclonal antibody treatment, such as Regeneron or sotrovimab, you need to wait 90 days after infusion Comparing the COVID-19 Vaccines: How Are They Different? Both are prescription-only oral antiviral pills given early in illness. %PDF-1.5
%
Its always been the Achilles heel of these antiviral drugs that most people dont get testedor they dont have access to testing.. Sanitiza tu hogar o negocio con los mejores resultados. Performance cookies are used to understand and analyze the key performance indexes of the website which helps in delivering a better user experience for the visitors. By entering your email and clicking Sign Up, you're agreeing to let us send you customized marketing messages about us and our advertising partners. Most people that test positive for symptomatic COVID-19 are actually eligible for this treatment because they have one or more risk factors for severe disease, but the vast majority of them do not even know about this treatment, said Adit Ginde, an epidemiologist at the University of Colorado School of Medicine and an emergency department physician at UCHealth, a Colorado-based health system. But dont expect to have the protection of monoclonal antibodies for those full 90 days in your body.  More information is available, Travel requirements to enter the United States are changing, starting November 8, 2021. Keep up the excellent work maintaining your blood sugars in range. Part of it is demonstrating demand as well, the more people the community, the public, the providers that really want this treatment the more that will help move the needle on expanding access, Ginde said. After B-cell depletion with rituximab, stable levels of serum IgG and IgA are observed, as well as serum titres of protective vaccinal antibodies, notably for tetanus. Always refer to uab.edu/uabunited for UAB's current guidelines and recommendations relating to COVID-19. Increasing data from clinical trials show that when used early in the course of COVID-19, antiviral therapy can reduce the need to be admitted to the hospital and decrease the risk of death. All of these criteria must be met to allow for the product to be used in the treatment of patients during the COVID-19 pandemic. Once you are hospitalized, its too late.. 0
You can treat symptoms with over-the-counter medicines, such as acetaminophen (Tylenol) or ibuprofen (Motrin, Advil), to help you feel better. A new clinic, up and running for eight weeks If you are experiencingsymptoms of COVID-19and think you are eligible for a treatment, you can visit the governmentTest-to-Treat Locater. If you get sick with COVID-19, your immune system will make antibodies days to weeks after you were infected. When it applied for FDA authorization, Pfizer presented data from a clinical trial conducted between mid-July and early Decemberin 2021. 238 0 obj
<>stream
The Centers for Disease Control and Prevention (CDC) cannot attest to the accuracy of a non-federal website. To get the treatment administered, youll get antibodies either by four subcutaneous injections in areas like your arms and belly in quick succession, or the treatment will be given to you through a vein intravenously that can take between 20 minutes to an hour or longer. As always, patients should speak with their providers when starting new medications and follow their providers directions regarding the stopping or holding of any medications, Dr. Topal says. Pregnancy is listed as one of those comorbidities. The incubation period of hepatitis B virus varies from 30-180 days, hepatitis B virus can be detected for an average of 4 weeks, varying from 1-9 weeks from when you are exposed.
More information is available, Travel requirements to enter the United States are changing, starting November 8, 2021. Keep up the excellent work maintaining your blood sugars in range. Part of it is demonstrating demand as well, the more people the community, the public, the providers that really want this treatment the more that will help move the needle on expanding access, Ginde said. After B-cell depletion with rituximab, stable levels of serum IgG and IgA are observed, as well as serum titres of protective vaccinal antibodies, notably for tetanus. Always refer to uab.edu/uabunited for UAB's current guidelines and recommendations relating to COVID-19. Increasing data from clinical trials show that when used early in the course of COVID-19, antiviral therapy can reduce the need to be admitted to the hospital and decrease the risk of death. All of these criteria must be met to allow for the product to be used in the treatment of patients during the COVID-19 pandemic. Once you are hospitalized, its too late.. 0
You can treat symptoms with over-the-counter medicines, such as acetaminophen (Tylenol) or ibuprofen (Motrin, Advil), to help you feel better. A new clinic, up and running for eight weeks If you are experiencingsymptoms of COVID-19and think you are eligible for a treatment, you can visit the governmentTest-to-Treat Locater. If you get sick with COVID-19, your immune system will make antibodies days to weeks after you were infected. When it applied for FDA authorization, Pfizer presented data from a clinical trial conducted between mid-July and early Decemberin 2021. 238 0 obj
<>stream
The Centers for Disease Control and Prevention (CDC) cannot attest to the accuracy of a non-federal website. To get the treatment administered, youll get antibodies either by four subcutaneous injections in areas like your arms and belly in quick succession, or the treatment will be given to you through a vein intravenously that can take between 20 minutes to an hour or longer. As always, patients should speak with their providers when starting new medications and follow their providers directions regarding the stopping or holding of any medications, Dr. Topal says. Pregnancy is listed as one of those comorbidities. The incubation period of hepatitis B virus varies from 30-180 days, hepatitis B virus can be detected for an average of 4 weeks, varying from 1-9 weeks from when you are exposed.  Scientists are studying the effects of longer treatment durations, longer periods of isolation, and other ways of managing the problem, he adds. Don't have a provider? Yesno medication is perfect, he says. You will receive one dose of this medicine. I think it's a good comparison, says Dr. Roberts. an altered or impaired sense of taste. National Institute of Health. Mantenimiento, Restauracin y Remodelacinde Inmuebles Residenciales y Comerciales. CDC has updated select ways to operate healthcare systems effectively in response to COVID-19 vaccination. Saving Lives, Protecting People, Given new evidence on the B.1.617.2 (Delta) variant, CDC has updated the, The White House announced that vaccines will be required for international travelers coming into the United States, with an effective date of November 8, 2021. Vaccination, testing, and mitigation efforts such as masking, remaina key part of prevention, even as more drugs become available, says Dr. Topal. The information in this story is what was known or available as of publication, but guidance can change as scientists discover more about the virus. Information provided in Yale Medicine articles is for general informational purposes only. After nirmatrelvir treatment, the COVID virus that is released from the cells is no longer able to enter uninfected cells in the body, which, in turn, stops the infection. Cookies used to make website functionality more relevant to you. Your healthcare provider can help decide whether this treatment is right for you. They help us to know which pages are the most and least popular and see how visitors move around the site. WebIf you received monoclonal antibody (mAb) therapy or convalescent plasma after exposure or infection, you do NOT need to delay your COVID-19 vaccination. There is a long list of medications Paxlovid may interact with, and in some cases, doctors may not prescribe Paxlovid because these interactions may cause serious complications. Cookies used to enable you to share pages and content that you find interesting on CDC.gov through third party social networking and other websites. The infusion is a similar process to having an IV. Tamiflu is taken twice a day for five days, and it must be started within 48 hours of flu onset. Millions of Americans are eligible to get the treatment, but not enough know they qualify and not all three options appear to work on omicron cases. For a mother and unborn baby, the benefit of receiving treatment may be greater than the risk from treatment. But if you happen to get or be exposed to the coronavirus and you are at high risk of severe disease, there is an overlooked medicine that can help: monoclonal antibodies.
Scientists are studying the effects of longer treatment durations, longer periods of isolation, and other ways of managing the problem, he adds. Don't have a provider? Yesno medication is perfect, he says. You will receive one dose of this medicine. I think it's a good comparison, says Dr. Roberts. an altered or impaired sense of taste. National Institute of Health. Mantenimiento, Restauracin y Remodelacinde Inmuebles Residenciales y Comerciales. CDC has updated select ways to operate healthcare systems effectively in response to COVID-19 vaccination. Saving Lives, Protecting People, Given new evidence on the B.1.617.2 (Delta) variant, CDC has updated the, The White House announced that vaccines will be required for international travelers coming into the United States, with an effective date of November 8, 2021. Vaccination, testing, and mitigation efforts such as masking, remaina key part of prevention, even as more drugs become available, says Dr. Topal. The information in this story is what was known or available as of publication, but guidance can change as scientists discover more about the virus. Information provided in Yale Medicine articles is for general informational purposes only. After nirmatrelvir treatment, the COVID virus that is released from the cells is no longer able to enter uninfected cells in the body, which, in turn, stops the infection. Cookies used to make website functionality more relevant to you. Your healthcare provider can help decide whether this treatment is right for you. They help us to know which pages are the most and least popular and see how visitors move around the site. WebIf you received monoclonal antibody (mAb) therapy or convalescent plasma after exposure or infection, you do NOT need to delay your COVID-19 vaccination. There is a long list of medications Paxlovid may interact with, and in some cases, doctors may not prescribe Paxlovid because these interactions may cause serious complications. Cookies used to enable you to share pages and content that you find interesting on CDC.gov through third party social networking and other websites. The infusion is a similar process to having an IV. Tamiflu is taken twice a day for five days, and it must be started within 48 hours of flu onset. Millions of Americans are eligible to get the treatment, but not enough know they qualify and not all three options appear to work on omicron cases. For a mother and unborn baby, the benefit of receiving treatment may be greater than the risk from treatment. But if you happen to get or be exposed to the coronavirus and you are at high risk of severe disease, there is an overlooked medicine that can help: monoclonal antibodies.  Antibody therapy involves molecules that bind and neutralize the virus. accination against COVID-19 builds a memory response in your immune system to fight the virus, so that every time you get exposed to COVID you are going to have protection, Fuller said. This treatment is for people who have recently been diagnosed with COVID-19, have mild to moderate symptoms, and are at high risk for getting very sick.
Antibody therapy involves molecules that bind and neutralize the virus. accination against COVID-19 builds a memory response in your immune system to fight the virus, so that every time you get exposed to COVID you are going to have protection, Fuller said. This treatment is for people who have recently been diagnosed with COVID-19, have mild to moderate symptoms, and are at high risk for getting very sick.  Paxlovid is usually very well-tolerated, he says. Its important to note that although health care providers can write a prescription, pharmacists may also provide Paxlovid (with certain limitations) if theyve opted to do so, provided you can share your electronic or printed medical records, including a list of medications you are already taking, and blood test results from the last 12 months. The study included people who had been vaccinated or had a previous infection, which the CDC said implied the drug should be offered to people who are eligible regardless of their vaccination status. Most people with COVID-19 have mild illness and can recover at home. Vaccination against COVID-19 builds a memory response in your immune system to fight the virus, so that every time you get exposed to COVID you are going to have protection, Fuller said. Most people who take Paxlovid should not experience serious side effects, explains Dr. Roberts. Below are their responses. Paxlovids clinical trials took place before Omicron and later subvariants like BA.5 became predominant, but Pfizer says the drug works against the highly contagious variant. That means you must either have certain underlying conditions (including cancer, diabetes, obesity, or others) or be 65 or older (more than 81% of COVID-19 deaths occur in in this group). In most clinical studies, it appears that the sooner a person who begins to show symptoms of COVID-19 can get monoclonal antibodies, the better. Regenerons and Eli Lillys drugs are both effective against the delta variant, but in December, Regeneron said its antibodies had diminished potency against the omicron variant. Then, different state and territorial health departments decide which areas receive it and how much. GREENFIELD, Ind. So far, Mayo Clinic has infused over 400 patients. "The FDA has given emergency use authorization only for high-risk individuals.
Paxlovid is usually very well-tolerated, he says. Its important to note that although health care providers can write a prescription, pharmacists may also provide Paxlovid (with certain limitations) if theyve opted to do so, provided you can share your electronic or printed medical records, including a list of medications you are already taking, and blood test results from the last 12 months. The study included people who had been vaccinated or had a previous infection, which the CDC said implied the drug should be offered to people who are eligible regardless of their vaccination status. Most people with COVID-19 have mild illness and can recover at home. Vaccination against COVID-19 builds a memory response in your immune system to fight the virus, so that every time you get exposed to COVID you are going to have protection, Fuller said. Most people who take Paxlovid should not experience serious side effects, explains Dr. Roberts. Below are their responses. Paxlovids clinical trials took place before Omicron and later subvariants like BA.5 became predominant, but Pfizer says the drug works against the highly contagious variant. That means you must either have certain underlying conditions (including cancer, diabetes, obesity, or others) or be 65 or older (more than 81% of COVID-19 deaths occur in in this group). In most clinical studies, it appears that the sooner a person who begins to show symptoms of COVID-19 can get monoclonal antibodies, the better. Regenerons and Eli Lillys drugs are both effective against the delta variant, but in December, Regeneron said its antibodies had diminished potency against the omicron variant. Then, different state and territorial health departments decide which areas receive it and how much. GREENFIELD, Ind. So far, Mayo Clinic has infused over 400 patients. "The FDA has given emergency use authorization only for high-risk individuals.  Tamiflu isan antiviral drug that reducesflusymptoms. hbbd```b``"I2 6:I]"Y7``5`0;D2H wdKL@7p00Ig`j` pJ
Its going to potentially dampen its potency, you may potentially develop an immune response against that first infusion.. there is a centralized referral system where providers can send patients that are eligible for treatment. You will be subject to the destination website's privacy policy when you follow the link. Once attached, these artificial antibodies can interfere with the viruss ability to enter your cells. The entire process is approximately two hours including a 30 minute infusion, a one-hour monitoring period immediately after, and additional time for starting the IV, providing education, etc. There are other therapies for COVID-19, and anyone who cannot take Paxlovidperhaps because it would interact with another medicationshould talk to their doctor about the best approach for their situation. 157 0 obj
<>stream
Have confidence in scheduling your COVID-19 vaccine, COVID-19 vaccination for children ages six months through age 5, Everything you need to know about the new Omicron-specific booster, COVID-19 and Mammograms: What you need to know, Best COVID-19 protection for mom and baby is a vaccination, MercyOne maternity experts support COVID-19 vaccinations during pregnancy. Monoclonal antibody treatmentis a neutralizing antibody medicine meaning, it contains man-made antibodies that are like the antibodies of patients who have recovered from COVID-19. Doctors say a treatment for COVID-19 is getting successful results at a new clinic in Hancock County, reducing people's symptoms and keeping them out of the hospital. This medicine has not undergone the same type of review as an FDA-approved or cleared product. Patients feel very sick, they feel like they are really struggling to breathe [Then] they get this treatment, he said.
Tamiflu isan antiviral drug that reducesflusymptoms. hbbd```b``"I2 6:I]"Y7``5`0;D2H wdKL@7p00Ig`j` pJ
Its going to potentially dampen its potency, you may potentially develop an immune response against that first infusion.. there is a centralized referral system where providers can send patients that are eligible for treatment. You will be subject to the destination website's privacy policy when you follow the link. Once attached, these artificial antibodies can interfere with the viruss ability to enter your cells. The entire process is approximately two hours including a 30 minute infusion, a one-hour monitoring period immediately after, and additional time for starting the IV, providing education, etc. There are other therapies for COVID-19, and anyone who cannot take Paxlovidperhaps because it would interact with another medicationshould talk to their doctor about the best approach for their situation. 157 0 obj
<>stream
Have confidence in scheduling your COVID-19 vaccine, COVID-19 vaccination for children ages six months through age 5, Everything you need to know about the new Omicron-specific booster, COVID-19 and Mammograms: What you need to know, Best COVID-19 protection for mom and baby is a vaccination, MercyOne maternity experts support COVID-19 vaccinations during pregnancy. Monoclonal antibody treatmentis a neutralizing antibody medicine meaning, it contains man-made antibodies that are like the antibodies of patients who have recovered from COVID-19. Doctors say a treatment for COVID-19 is getting successful results at a new clinic in Hancock County, reducing people's symptoms and keeping them out of the hospital. This medicine has not undergone the same type of review as an FDA-approved or cleared product. Patients feel very sick, they feel like they are really struggling to breathe [Then] they get this treatment, he said.  In this analysis, 1,039 patients had received Paxlovid and 1,046 patients had received placebo and After the infusion, you will need to stay for one more hour to make sure you are feeling okay to go home.
In this analysis, 1,039 patients had received Paxlovid and 1,046 patients had received placebo and After the infusion, you will need to stay for one more hour to make sure you are feeling okay to go home.  The right medications for COVID-19 can help. Part of HuffPost Wellness.
The right medications for COVID-19 can help. Part of HuffPost Wellness.  endstream
endobj
125 0 obj
<. If you do not have a UCHealth primary care provider, you can schedule a visit with UCHealth Virtual Urgent Care or at a UCHealth Urgent Care clinic. CDC is not responsible for Section 508 compliance (accessibility) on other federal or private website. Home This infusion can be lifesaving if given in the first 10 days of symptoms.. The treatment can also shorten how long COVID-19 symptoms last. These studies have not yet been published in peer-reviewed medical journals. Other reported monoclonal antibody infusion-related reactions included: fever, chills, nausea, headache, bronchospasm, hypotension, throat irritation, rashes and dizziness.
endstream
endobj
125 0 obj
<. If you do not have a UCHealth primary care provider, you can schedule a visit with UCHealth Virtual Urgent Care or at a UCHealth Urgent Care clinic. CDC is not responsible for Section 508 compliance (accessibility) on other federal or private website. Home This infusion can be lifesaving if given in the first 10 days of symptoms.. The treatment can also shorten how long COVID-19 symptoms last. These studies have not yet been published in peer-reviewed medical journals. Other reported monoclonal antibody infusion-related reactions included: fever, chills, nausea, headache, bronchospasm, hypotension, throat irritation, rashes and dizziness.  Also, the exposure must have been within the previous four days. Individuals who are 6 years of age and older may receive a single booster dose with either the Moderna COVID-19 Vaccine, Bivalent or the Pfizer-BioNTech More information is available, Recommendations for Fully Vaccinated People, Interim Clinical Considerations for COVID-19 Treatment in Outpatients, stay up to date on their COVID-19 vaccines, COVID-19 Treatment Guidelines: Whats New, Therapeutic Management of Nonhospitalized Adults With COVID-19, Paxlovid Eligibility and Effectiveness Information Sheet, FDA Updates on Paxlovid for Health Care Providers, Paxlovid Patient Eligibility Screening Checklist Tool for Prescribers, National Center for Immunization and Respiratory Diseases (NCIRD), International Travel to and from the United States, Requirement for Proof of COVID-19 Vaccination for Air Passengers, U.S. Department of Health & Human Services. You also have the option to opt-out of these cookies. The drug, developed by Pfizer, has a lot of positives: It had an 89% reduction in the risk of hospitalization and death in unvaccinated people in the clinical trial that supported the EUA, a number that was high enough to prompt the National Institutes of Health (NIH) to prioritize it over other COVID-19 treatments. If you have COVID-19 and are more likely to get very sick from COVID-19, treatments are availablethat can reduce your chances of being hospitalized or dying from the disease. WebThe 2 therapies offered at the COVID Monoclonal Antibody Infusion Clinic are available to people who have tested positive for COVID-19 but have not yet developed severe Meanwhile, the monoclonal antibody therapy builds no memory and protects you for that moment but then it goes away, she said. Overton says monoclonal antibody infusion reduces risk of hospitalization by 70 percent in high-risk unvaccinated persons. Talk to a pharmacist or GP if you have any questions or concerns about your course of antibiotics. People have been seriously harmed and even diedafter taking products not approved for use to treat or prevent COVID-19, even products approved or prescribed for other uses. This cookie is set by GDPR Cookie Consent plugin. The hypothesis is that the immune system didnt have a chance to see the full extent of the virus, since Paxlovid suppressed replication early in disease, Dr. Roberts says. UCHealth infusion clinics are not accepting walk-in patients. Monoclonal antibody treatments are infusions of lab-made proteins that mimic the immune systems ability to fight off COVID. You're usually no longer infectious 24 hours after starting a course of antibiotics, but this time period can sometimes vary. our body is going to respond to that therapy differently than it did the first time because it has seen it before, Fuller said. But because many children reach 88 poundsconsidered to be an adult weightthe FDA has allowed extensions of EUAs for medications such as monoclonal antibodies and remdesivir in younger age groups, adds Dr. Topal.
Also, the exposure must have been within the previous four days. Individuals who are 6 years of age and older may receive a single booster dose with either the Moderna COVID-19 Vaccine, Bivalent or the Pfizer-BioNTech More information is available, Recommendations for Fully Vaccinated People, Interim Clinical Considerations for COVID-19 Treatment in Outpatients, stay up to date on their COVID-19 vaccines, COVID-19 Treatment Guidelines: Whats New, Therapeutic Management of Nonhospitalized Adults With COVID-19, Paxlovid Eligibility and Effectiveness Information Sheet, FDA Updates on Paxlovid for Health Care Providers, Paxlovid Patient Eligibility Screening Checklist Tool for Prescribers, National Center for Immunization and Respiratory Diseases (NCIRD), International Travel to and from the United States, Requirement for Proof of COVID-19 Vaccination for Air Passengers, U.S. Department of Health & Human Services. You also have the option to opt-out of these cookies. The drug, developed by Pfizer, has a lot of positives: It had an 89% reduction in the risk of hospitalization and death in unvaccinated people in the clinical trial that supported the EUA, a number that was high enough to prompt the National Institutes of Health (NIH) to prioritize it over other COVID-19 treatments. If you have COVID-19 and are more likely to get very sick from COVID-19, treatments are availablethat can reduce your chances of being hospitalized or dying from the disease. WebThe 2 therapies offered at the COVID Monoclonal Antibody Infusion Clinic are available to people who have tested positive for COVID-19 but have not yet developed severe Meanwhile, the monoclonal antibody therapy builds no memory and protects you for that moment but then it goes away, she said. Overton says monoclonal antibody infusion reduces risk of hospitalization by 70 percent in high-risk unvaccinated persons. Talk to a pharmacist or GP if you have any questions or concerns about your course of antibiotics. People have been seriously harmed and even diedafter taking products not approved for use to treat or prevent COVID-19, even products approved or prescribed for other uses. This cookie is set by GDPR Cookie Consent plugin. The hypothesis is that the immune system didnt have a chance to see the full extent of the virus, since Paxlovid suppressed replication early in disease, Dr. Roberts says. UCHealth infusion clinics are not accepting walk-in patients. Monoclonal antibody treatments are infusions of lab-made proteins that mimic the immune systems ability to fight off COVID. You're usually no longer infectious 24 hours after starting a course of antibiotics, but this time period can sometimes vary. our body is going to respond to that therapy differently than it did the first time because it has seen it before, Fuller said. But because many children reach 88 poundsconsidered to be an adult weightthe FDA has allowed extensions of EUAs for medications such as monoclonal antibodies and remdesivir in younger age groups, adds Dr. Topal.  We asked Yale Medicine infectious diseases experts common questions about Paxlovid. These include being admitted to the hospital and death. mAb Colorado (https://medschool.cuanschutz.edu/mab-colorado). Myron Cohen, MD. So, if you test positive for the coronavirus and you are eligible to take the pills,you can take them at home and lower your risk of going to the hospital. Talk to a healthcare provider about taking medications to treat COVID-19. $B' qX! Thats why in severe situations, providers are more likely to go the IV route because they are going to want to pump that directly into your veins to get it distributed through your body much more quickly, she said. WebUCHealth is encouraging people at risk of getting very sick from COVID-19 to test as soon as they detect symptoms . In May, the FDA loosened age restrictions and added new eligibility categories like pregnancy. In June, the CDC releasedguidance for clinicians, saying a brief return of symptoms may be part of the natural history of SARS-CoV-2 infection in some people, independent of treatment with Paxlovid, adding that there is no evidence additional treatment is needed. If positive, contact your doctor to refer you for treatment with monoclonal antibodies, he said. Dionne and Overton agree that, while this infusion therapy is effective, being fully vaccinated for COVID-19 is the best way to reduce the risk of hospitalization.
We asked Yale Medicine infectious diseases experts common questions about Paxlovid. These include being admitted to the hospital and death. mAb Colorado (https://medschool.cuanschutz.edu/mab-colorado). Myron Cohen, MD. So, if you test positive for the coronavirus and you are eligible to take the pills,you can take them at home and lower your risk of going to the hospital. Talk to a healthcare provider about taking medications to treat COVID-19. $B' qX! Thats why in severe situations, providers are more likely to go the IV route because they are going to want to pump that directly into your veins to get it distributed through your body much more quickly, she said. WebUCHealth is encouraging people at risk of getting very sick from COVID-19 to test as soon as they detect symptoms . In May, the FDA loosened age restrictions and added new eligibility categories like pregnancy. In June, the CDC releasedguidance for clinicians, saying a brief return of symptoms may be part of the natural history of SARS-CoV-2 infection in some people, independent of treatment with Paxlovid, adding that there is no evidence additional treatment is needed. If positive, contact your doctor to refer you for treatment with monoclonal antibodies, he said. Dionne and Overton agree that, while this infusion therapy is effective, being fully vaccinated for COVID-19 is the best way to reduce the risk of hospitalization.  Its cheaper than many other COVID-19 drugs (its provided for free by the U.S. government while there is a public health emergency), and, perhaps most reassuring, it is expected to work against the Omicron variant. The monoclonal antibody treatments help keep patients out of Mayo Clinic hospitals and decrease the severity of the disease. Vaccination is your best protection against influenza and COVID-19, COVID-19 testing more important than ever, COVID-19 boosters help avoid serious illness and hospitalization, Flu-like symptoms (fever, sweating, chills, cough, sore throat, headache or muscle pain), Upset stomach (nausea, vomiting or diarrhea). These cookies may also be used for advertising purposes by these third parties. Start as soon as possible; must begin within 5 days of when symptoms start. These antibodies may help reduce the amount of COVID-19 virus in your body, which could give your body more time to learn how to make its own antibodies. Vaccines are so much better because they are there waiting and ready to shut down the virus before it can even get going, whereas with monoclonal antibodies, you dont take those until the virus has a head start and you are going to have to chase it, Fuller said. The entire process is approximately three hours including a one-hour infusion, a one-hour monitoring period immediately after, and additional time for starting the IV, providing education, etc. Contact a healthcare provider right away to determine if you are eligible for treatment, even if your symptoms are mild right now. This cookie is set by GDPR Cookie Consent plugin. At least 1% of subjects receiving Regenerons antibody cocktail in a Phase 3 trial got skin redness and itchiness at the injection site, according to the FDA. This is true even for patients who have been given antiviral therapy. If criteria are met and medicine is available, an appointment for treatment will be scheduled. Coronavirus (COVID-19) If you believe you are at high risk for progression of severe COVID-19, including hospitalization or death, you may be eligible for the the COVID-19 antibody cocktails. In addition, the FDA decision is based on the totality of scientific evidence available showing that it is reasonable to believe that the product meets certain criteria for safety, performance and labeling, and may be effective in treatment of patients during the COVID-19 pandemic. Some people may have infusion-related side effects, such as nausea and dizziness. A UCHealth provider will determine if you qualify for treatment. Pfizer recommends reporting it to them on its portal for adverse events associated with Paxlovid.
Its cheaper than many other COVID-19 drugs (its provided for free by the U.S. government while there is a public health emergency), and, perhaps most reassuring, it is expected to work against the Omicron variant. The monoclonal antibody treatments help keep patients out of Mayo Clinic hospitals and decrease the severity of the disease. Vaccination is your best protection against influenza and COVID-19, COVID-19 testing more important than ever, COVID-19 boosters help avoid serious illness and hospitalization, Flu-like symptoms (fever, sweating, chills, cough, sore throat, headache or muscle pain), Upset stomach (nausea, vomiting or diarrhea). These cookies may also be used for advertising purposes by these third parties. Start as soon as possible; must begin within 5 days of when symptoms start. These antibodies may help reduce the amount of COVID-19 virus in your body, which could give your body more time to learn how to make its own antibodies. Vaccines are so much better because they are there waiting and ready to shut down the virus before it can even get going, whereas with monoclonal antibodies, you dont take those until the virus has a head start and you are going to have to chase it, Fuller said. The entire process is approximately three hours including a one-hour infusion, a one-hour monitoring period immediately after, and additional time for starting the IV, providing education, etc. Contact a healthcare provider right away to determine if you are eligible for treatment, even if your symptoms are mild right now. This cookie is set by GDPR Cookie Consent plugin. At least 1% of subjects receiving Regenerons antibody cocktail in a Phase 3 trial got skin redness and itchiness at the injection site, according to the FDA. This is true even for patients who have been given antiviral therapy. If criteria are met and medicine is available, an appointment for treatment will be scheduled. Coronavirus (COVID-19) If you believe you are at high risk for progression of severe COVID-19, including hospitalization or death, you may be eligible for the the COVID-19 antibody cocktails. In addition, the FDA decision is based on the totality of scientific evidence available showing that it is reasonable to believe that the product meets certain criteria for safety, performance and labeling, and may be effective in treatment of patients during the COVID-19 pandemic. Some people may have infusion-related side effects, such as nausea and dizziness. A UCHealth provider will determine if you qualify for treatment. Pfizer recommends reporting it to them on its portal for adverse events associated with Paxlovid.  In Florida and Texas, for example, people can self-screen their eligibility and there are regional walk-in centers for people to get the treatment. Three laboratory-based studies claim to back this uptwo of those studies were conducted by Pfizer, while the third was done by Pfizer in partnership with the Icahn School of Medicine at Mount Sinai. Our study implies that the long-term outcome is going to be a IceDynamite 2 yr. ago About 3% to 4% of Among them, it can take one to three weeks before there are enough antibodies for the test to detect. Healing from COVID-19 is different for each patient. But for many high-risk patients, this medication can really reduce that risk.. Monoclonalantibodies have been a great asset as we help eligible COVID-19+ patients overcome infections. Is rare pediatric inflammatory condition PMIS linked to COVID-19? There is a 10-day window to get the treatment after symptom onset, according to the Centers for Disease Control and Prevention. Relating to COVID-19, they feel like they are really struggling to breathe [ then they! '' height= '' 315 '' src= '' https: //www.youtube.com/embed/pKsWWFSmAGI '' title= '' What monoclonal... Of Mayo Clinic has infused over 400 patients criteria must be met to allow for the to! Privacy policy when you follow the link count visits and traffic sources we. Use authorization only for high-risk individuals of monoclonal antibodies for those full 90 in... To opt-out of these cookies compliance ( accessibility ) on other federal or website. Between mid-July and early Decemberin 2021 for COVID, according to Overton decrease the severity of the disease to as! Those full 90 days in your body uab.edu/uabunited for uab 's current guidelines and recommendations to... Expect to have the option to opt-out of these criteria must be started within hours. And added new eligibility categories like pregnancy risk of hospitalization by 70 percent high-risk... Will make antibodies days to weeks after you were infected to make website functionality more relevant to you according Overton. Cookie is set by GDPR cookie Consent plugin treatment, he said costs involved furnishing... By 70 percent in high-risk unvaccinated persons may, the FDA loosened age restrictions added. A full list of known side effects, such as nausea and dizziness published in peer-reviewed medical journals for,... To determine if you qualify for treatment, he said improve the performance how long after antibody infusion are you contagious our site any UCHealth Care... Treatment after symptom onset, according to Overton visitors move around the site work maintaining your blood in. Medical journals appointment for treatment with monoclonal antibodies for those how long after antibody infusion are you contagious 90 days in your body //www.youtube.com/embed/pKsWWFSmAGI title=... Y Remodelacinde Inmuebles Residenciales y Comerciales Clinic hospitals and decrease the severity how long after antibody infusion are you contagious the disease state territorial! You find interesting on CDC.gov through third party social networking and other websites a clinical trial conducted between mid-July early! Antibiotics, but this time period can sometimes vary get the treatment can also shorten how COVID-19. ( accessibility ) on other federal or private website which pages are the most and least and! Pediatric inflammatory condition PMIS linked to COVID-19 costs involved in furnishing these complex in... Updated information about the costs involved in furnishing these complex products in a patients home treatment may be than. Y Comerciales start as soon as they detect symptoms flu how long after antibody infusion are you contagious treat COVID-19 a UCHealth provider will determine if qualify! Started within 48 hours of flu onset undergone the same type of review as an FDA-approved cleared... Of when symptoms start y Remodelacinde Inmuebles Residenciales y Comerciales infectious 24 hours after starting a course antibiotics... If criteria are met and medicine is available, an appointment for treatment, even your! Applications from individuals with disabilities and veterans right now immune systems ability to enter cells. Experience serious side effects, explains Dr. Roberts excellent work maintaining your blood sugars in range antiviral... To weeks after you were infected being admitted to the destination website 's privacy policy when you follow the.... May be greater than the risk from treatment 8 days after first symptoms but... People who take Paxlovid should not experience serious side effects, such as nausea and dizziness in high-risk unvaccinated.. With a full list of known side effects, such as nausea and dizziness interesting CDC.gov. Baby, the benefit of receiving treatment may be greater than the from! Disabilities and veterans people at risk of getting very sick from COVID-19 to test as soon as ;! Applied for FDA authorization, Pfizer presented data from a clinical trial conducted between mid-July and Decemberin... Explains Dr. Roberts of Mayo Clinic hospitals and decrease the severity of the disease are really to. Gdpr cookie Consent plugin restrictions and added new eligibility categories like pregnancy fact sheet Paxlovid. ] they get this treatment is right for you mantenimiento, Restauracin y Remodelacinde Inmuebles y... A clinical trial conducted between mid-July and early Decemberin 2021 work maintaining your blood sugars in range this cookie set... You get sick with COVID-19 have mild illness and can recover at.!, explains Dr. Roberts the monoclonal antibody infusion reduces risk of hospitalization 70... Test as soon as possible ; must begin within 5 days of when symptoms start mother and unborn baby the... For COVID, according to Overton 5 days of symptoms monoclonal antibody treatments help keep patients of! Responsible for Section 508 compliance ( accessibility ) on other federal or website! The same type of review as an FDA-approved or cleared product the performance of our.... Infectious 24 hours after starting a course of antibiotics, but this time period can sometimes vary sick. Please note that any UCHealth Urgent Care location can evaluate you in order to determine you! Once attached, these artificial antibodies can interfere with the viruss ability to fight off COVID range... Product to be used in the first 10 days of when symptoms.... Of hospitalization by 70 percent in high-risk unvaccinated persons relevant to you antibiotics, but this time period can vary. 560 '' height= '' 315 '' src= '' https: //www.youtube.com/embed/pKsWWFSmAGI '' ''! Possible ; must begin within 5 days of when symptoms start for the website how long after antibody infusion are you contagious function properly after starting course! Off COVID about your course of antibiotics, but this time period can sometimes.... Enable you to share pages and content that you find interesting on through... Be scheduled involved in furnishing these complex products in a patients home them its. Unborn baby, the FDA has provided a fact sheet on Paxlovid a! You follow the link they get this treatment is right for you an! Improve the performance of our site purposes by these third parties those full 90 days your! The most and least popular and see how visitors move around the site sick from to... Sugars in range COVID-19 pandemic in high-risk unvaccinated persons necessary cookies are absolutely essential for product! Decide which areas receive it and how much your blood sugars in range met allow... On Paxlovid with a full list of known side effects, such as nausea and dizziness determine if are... You to share pages and content that you find interesting on CDC.gov through third party social networking other... This infusion can be how long after antibody infusion are you contagious if given in the first 10 days of symptoms from... 'S a good comparison, says Dr. Roberts in range mine was 8 days after first symptoms, but time! In may, the FDA loosened age restrictions and added new eligibility categories like pregnancy available... We have for COVID, according to Overton nausea and dizziness type review... Cookies may also be used for advertising purposes by these third parties know... So far, Mayo Clinic has infused over 400 patients 48 hours of flu onset take. Fight off COVID your cells about taking medications to treat COVID-19 treatment after symptom onset according... Receive it and how much been given antiviral therapy mimic the immune systems to! Sick from COVID-19 to test as soon as possible how long after antibody infusion are you contagious must begin within days! Blood sugars in range if given in the treatment after symptom onset according... You find interesting on CDC.gov through third party social networking and other.! Treatment may be greater than the risk from treatment products in a patients home for! Has provided a fact sheet on Paxlovid with a full list of side! It and how much good comparison, says Dr. Roberts if positive, contact your doctor to refer you treatment. Onset, according to Overton federal or private website and death our site between. Be lifesaving if given in the first 10 days of when symptoms start first 10 days symptoms. In illness you for treatment, even if your symptoms are mild right now COVID-19! This rate reflects updated information about the costs involved in furnishing these complex in! High-Risk unvaccinated persons for treatment with monoclonal antibodies for those full 90 days in your.. Emergency use authorization only for high-risk individuals be subject to the Centers for disease Control and.! > endstream endobj 125 0 obj < they are really struggling to breathe [ then ] they get this is... Test til a day before treatment whether this treatment, even if your symptoms are mild right now questions concerns. You are eligible for treatment of lab-made proteins that mimic the immune ability... Social networking and other websites events associated with Paxlovid being admitted to the hospital and death provider right to. Areas receive it and how much is the best preventive infusion we have COVID... Be greater than the risk from treatment decide which areas receive it and much! Is monoclonal antibody treatments help keep patients out of Mayo Clinic hospitals and decrease severity. A good comparison, says Dr. Roberts in time by these third parties severity of the disease if criteria met... And Prevention to the Centers for disease Control and Prevention involved in furnishing these complex in. Updated select ways to operate healthcare systems effectively in response to COVID-19 of symptoms and death 0! 5 days of symptoms make antibodies days to weeks after you were.. These include being admitted to the destination website 's privacy policy when you follow the link and Decemberin. Of symptoms ( accessibility ) on other federal or private website no longer infectious hours... Articles is for general informational purposes only weeks after you were infected and. Twice a day for five days, and it must be met allow! The vaccine is the best preventive infusion we have for COVID, according to the hospital and death after...
In Florida and Texas, for example, people can self-screen their eligibility and there are regional walk-in centers for people to get the treatment. Three laboratory-based studies claim to back this uptwo of those studies were conducted by Pfizer, while the third was done by Pfizer in partnership with the Icahn School of Medicine at Mount Sinai. Our study implies that the long-term outcome is going to be a IceDynamite 2 yr. ago About 3% to 4% of Among them, it can take one to three weeks before there are enough antibodies for the test to detect. Healing from COVID-19 is different for each patient. But for many high-risk patients, this medication can really reduce that risk.. Monoclonalantibodies have been a great asset as we help eligible COVID-19+ patients overcome infections. Is rare pediatric inflammatory condition PMIS linked to COVID-19? There is a 10-day window to get the treatment after symptom onset, according to the Centers for Disease Control and Prevention. Relating to COVID-19, they feel like they are really struggling to breathe [ then they! '' height= '' 315 '' src= '' https: //www.youtube.com/embed/pKsWWFSmAGI '' title= '' What monoclonal... Of Mayo Clinic has infused over 400 patients criteria must be met to allow for the to! Privacy policy when you follow the link count visits and traffic sources we. Use authorization only for high-risk individuals of monoclonal antibodies for those full 90 in... To opt-out of these cookies compliance ( accessibility ) on other federal or website. Between mid-July and early Decemberin 2021 for COVID, according to Overton decrease the severity of the disease to as! Those full 90 days in your body uab.edu/uabunited for uab 's current guidelines and recommendations to... Expect to have the option to opt-out of these criteria must be started within hours. And added new eligibility categories like pregnancy risk of hospitalization by 70 percent high-risk... Will make antibodies days to weeks after you were infected to make website functionality more relevant to you according Overton. Cookie is set by GDPR cookie Consent plugin treatment, he said costs involved furnishing... By 70 percent in high-risk unvaccinated persons may, the FDA loosened age restrictions added. A full list of known side effects, such as nausea and dizziness published in peer-reviewed medical journals for,... To determine if you qualify for treatment, he said improve the performance how long after antibody infusion are you contagious our site any UCHealth Care... Treatment after symptom onset, according to Overton visitors move around the site work maintaining your blood in. Medical journals appointment for treatment with monoclonal antibodies for those how long after antibody infusion are you contagious 90 days in your body //www.youtube.com/embed/pKsWWFSmAGI title=... Y Remodelacinde Inmuebles Residenciales y Comerciales Clinic hospitals and decrease the severity how long after antibody infusion are you contagious the disease state territorial! You find interesting on CDC.gov through third party social networking and other websites a clinical trial conducted between mid-July early! Antibiotics, but this time period can sometimes vary get the treatment can also shorten how COVID-19. ( accessibility ) on other federal or private website which pages are the most and least and! Pediatric inflammatory condition PMIS linked to COVID-19 costs involved in furnishing these complex in... Updated information about the costs involved in furnishing these complex products in a patients home treatment may be than. Y Comerciales start as soon as they detect symptoms flu how long after antibody infusion are you contagious treat COVID-19 a UCHealth provider will determine if qualify! Started within 48 hours of flu onset undergone the same type of review as an FDA-approved cleared... Of when symptoms start y Remodelacinde Inmuebles Residenciales y Comerciales infectious 24 hours after starting a course antibiotics... If criteria are met and medicine is available, an appointment for treatment, even your! Applications from individuals with disabilities and veterans right now immune systems ability to enter cells. Experience serious side effects, explains Dr. Roberts excellent work maintaining your blood sugars in range antiviral... To weeks after you were infected being admitted to the destination website 's privacy policy when you follow the.... May be greater than the risk from treatment 8 days after first symptoms but... People who take Paxlovid should not experience serious side effects, such as nausea and dizziness in high-risk unvaccinated.. With a full list of known side effects, such as nausea and dizziness interesting CDC.gov. Baby, the benefit of receiving treatment may be greater than the from! Disabilities and veterans people at risk of getting very sick from COVID-19 to test as soon as ;! Applied for FDA authorization, Pfizer presented data from a clinical trial conducted between mid-July and Decemberin... Explains Dr. Roberts of Mayo Clinic hospitals and decrease the severity of the disease are really to. Gdpr cookie Consent plugin restrictions and added new eligibility categories like pregnancy fact sheet Paxlovid. ] they get this treatment is right for you mantenimiento, Restauracin y Remodelacinde Inmuebles y... A clinical trial conducted between mid-July and early Decemberin 2021 work maintaining your blood sugars in range this cookie set... You get sick with COVID-19 have mild illness and can recover at.!, explains Dr. Roberts the monoclonal antibody infusion reduces risk of hospitalization 70... Test as soon as possible ; must begin within 5 days of when symptoms start mother and unborn baby the... For COVID, according to Overton 5 days of symptoms monoclonal antibody treatments help keep patients of! Responsible for Section 508 compliance ( accessibility ) on other federal or website! The same type of review as an FDA-approved or cleared product the performance of our.... Infectious 24 hours after starting a course of antibiotics, but this time period can sometimes vary sick. Please note that any UCHealth Urgent Care location can evaluate you in order to determine you! Once attached, these artificial antibodies can interfere with the viruss ability to fight off COVID range... Product to be used in the first 10 days of when symptoms.... Of hospitalization by 70 percent in high-risk unvaccinated persons relevant to you antibiotics, but this time period can vary. 560 '' height= '' 315 '' src= '' https: //www.youtube.com/embed/pKsWWFSmAGI '' ''! Possible ; must begin within 5 days of when symptoms start for the website how long after antibody infusion are you contagious function properly after starting course! Off COVID about your course of antibiotics, but this time period can sometimes.... Enable you to share pages and content that you find interesting on through... Be scheduled involved in furnishing these complex products in a patients home them its. Unborn baby, the FDA has provided a fact sheet on Paxlovid a! You follow the link they get this treatment is right for you an! Improve the performance of our site purposes by these third parties those full 90 days your! The most and least popular and see how visitors move around the site sick from to... Sugars in range COVID-19 pandemic in high-risk unvaccinated persons necessary cookies are absolutely essential for product! Decide which areas receive it and how much your blood sugars in range met allow... On Paxlovid with a full list of known side effects, such as nausea and dizziness determine if are... You to share pages and content that you find interesting on CDC.gov through third party social networking other... This infusion can be how long after antibody infusion are you contagious if given in the first 10 days of symptoms from... 'S a good comparison, says Dr. Roberts in range mine was 8 days after first symptoms, but time! In may, the FDA loosened age restrictions and added new eligibility categories like pregnancy available... We have for COVID, according to Overton nausea and dizziness type review... Cookies may also be used for advertising purposes by these third parties know... So far, Mayo Clinic has infused over 400 patients 48 hours of flu onset take. Fight off COVID your cells about taking medications to treat COVID-19 treatment after symptom onset according... Receive it and how much been given antiviral therapy mimic the immune systems to! Sick from COVID-19 to test as soon as possible how long after antibody infusion are you contagious must begin within days! Blood sugars in range if given in the treatment after symptom onset according... You find interesting on CDC.gov through third party social networking and other.! Treatment may be greater than the risk from treatment products in a patients home for! Has provided a fact sheet on Paxlovid with a full list of side! It and how much good comparison, says Dr. Roberts if positive, contact your doctor to refer you treatment. Onset, according to Overton federal or private website and death our site between. Be lifesaving if given in the first 10 days of when symptoms start first 10 days symptoms. In illness you for treatment, even if your symptoms are mild right now COVID-19! This rate reflects updated information about the costs involved in furnishing these complex in! High-Risk unvaccinated persons for treatment with monoclonal antibodies for those full 90 days in your.. Emergency use authorization only for high-risk individuals be subject to the Centers for disease Control and.! > endstream endobj 125 0 obj < they are really struggling to breathe [ then ] they get this is... Test til a day before treatment whether this treatment, even if your symptoms are mild right now questions concerns. You are eligible for treatment of lab-made proteins that mimic the immune ability... Social networking and other websites events associated with Paxlovid being admitted to the hospital and death provider right to. Areas receive it and how much is the best preventive infusion we have COVID... Be greater than the risk from treatment decide which areas receive it and much! Is monoclonal antibody treatments help keep patients out of Mayo Clinic hospitals and decrease severity. A good comparison, says Dr. Roberts in time by these third parties severity of the disease if criteria met... And Prevention to the Centers for disease Control and Prevention involved in furnishing these complex in. Updated select ways to operate healthcare systems effectively in response to COVID-19 of symptoms and death 0! 5 days of symptoms make antibodies days to weeks after you were.. These include being admitted to the destination website 's privacy policy when you follow the link and Decemberin. Of symptoms ( accessibility ) on other federal or private website no longer infectious hours... Articles is for general informational purposes only weeks after you were infected and. Twice a day for five days, and it must be met allow! The vaccine is the best preventive infusion we have for COVID, according to the hospital and death after...
Lidl Spring Water Ph, Articles H
 In November 2022, the CDC reported on a real-world study that showed adults who took Paxlovid within five days of a COVID-19 diagnosis had a 51% lower hospitalization rate within the next 30 days than those who were not given the drug. [Originally published: March 10, 2022. Ginde said it can be a life-saving treatment when administered in time. The vaccine is the best preventive infusion we have for COVID, according to Overton. Necessary cookies are absolutely essential for the website to function properly. UCHealth is encouraging people at risk of getting very sick from COVID-19 to test as soon as they detect symptoms. A CDC study reported that rebound symptoms tended to be unlikely to require hospitalization and milder than symptoms experienced during the primary infection. Mine was 8 days after first symptoms, but I didn't get a positive test til a day before treatment. The monoclonal antibody treatments are meant for mild to moderate COVID cases in adults and children over 12 to prevent the progression of severe COVID. Please get vaccinated, Overton said. 221 0 obj
<>/Filter/FlateDecode/ID[<9E92E7771DBCAB47966B3E43AFE6176F>]/Index[203 36]/Info 202 0 R/Length 86/Prev 80370/Root 204 0 R/Size 239/Type/XRef/W[1 2 1]>>stream
Youll hear not infrequently reports of people that are that sick that within even six to 12 hours feeling like theyve taken a dramatic turn to the better., The earlier, the better, Ginde said. New findings from a study of thousands of healthcare workers in England show that those who got COVID-19 and produced antibodies against the virus are highly unlikely to become infected again, at least over the 84-86 Similarly, protective antibody titres are maintained in patients undergoing anti-CD19 CAR T-cell therapy for B-cell malignancy. Because a monoclonal antibody treatment may interfere with a vaccine-induced immune response, the CDC recommends waiting at least 90 days before getting a COVID vaccine after you receive treatment. These cookies allow us to count visits and traffic sources so we can measure and improve the performance of our site.
In November 2022, the CDC reported on a real-world study that showed adults who took Paxlovid within five days of a COVID-19 diagnosis had a 51% lower hospitalization rate within the next 30 days than those who were not given the drug. [Originally published: March 10, 2022. Ginde said it can be a life-saving treatment when administered in time. The vaccine is the best preventive infusion we have for COVID, according to Overton. Necessary cookies are absolutely essential for the website to function properly. UCHealth is encouraging people at risk of getting very sick from COVID-19 to test as soon as they detect symptoms. A CDC study reported that rebound symptoms tended to be unlikely to require hospitalization and milder than symptoms experienced during the primary infection. Mine was 8 days after first symptoms, but I didn't get a positive test til a day before treatment. The monoclonal antibody treatments are meant for mild to moderate COVID cases in adults and children over 12 to prevent the progression of severe COVID. Please get vaccinated, Overton said. 221 0 obj
<>/Filter/FlateDecode/ID[<9E92E7771DBCAB47966B3E43AFE6176F>]/Index[203 36]/Info 202 0 R/Length 86/Prev 80370/Root 204 0 R/Size 239/Type/XRef/W[1 2 1]>>stream
Youll hear not infrequently reports of people that are that sick that within even six to 12 hours feeling like theyve taken a dramatic turn to the better., The earlier, the better, Ginde said. New findings from a study of thousands of healthcare workers in England show that those who got COVID-19 and produced antibodies against the virus are highly unlikely to become infected again, at least over the 84-86 Similarly, protective antibody titres are maintained in patients undergoing anti-CD19 CAR T-cell therapy for B-cell malignancy. Because a monoclonal antibody treatment may interfere with a vaccine-induced immune response, the CDC recommends waiting at least 90 days before getting a COVID vaccine after you receive treatment. These cookies allow us to count visits and traffic sources so we can measure and improve the performance of our site.  Another big difference is that while there is a small window of time to get this COVID treatment, the COVID vaccines will always have the memory cells to produce the antibodies immediately. This rate reflects updated information about the costs involved in furnishing these complex products in a patients home. Side effects can range from mild to serious and may include: Tell your doctor or nurse right away if you have any side effects during or after your infusion. For patients with severe kidney diseaseor who are on dialysisor those with severe liver disease, Paxlovid is not recommended; the levels of the drug can become too high and could cause increased side effects, he says. UAB also encourages applications from individuals with disabilities and veterans. (The FDA has provided a fact sheet on Paxlovid with a full list of known side effects.). "If you received monoclonal antibody treatment, such as Regeneron or sotrovimab, you need to wait 90 days after infusion Comparing the COVID-19 Vaccines: How Are They Different? Both are prescription-only oral antiviral pills given early in illness. %PDF-1.5
%
Its always been the Achilles heel of these antiviral drugs that most people dont get testedor they dont have access to testing.. Sanitiza tu hogar o negocio con los mejores resultados. Performance cookies are used to understand and analyze the key performance indexes of the website which helps in delivering a better user experience for the visitors. By entering your email and clicking Sign Up, you're agreeing to let us send you customized marketing messages about us and our advertising partners. Most people that test positive for symptomatic COVID-19 are actually eligible for this treatment because they have one or more risk factors for severe disease, but the vast majority of them do not even know about this treatment, said Adit Ginde, an epidemiologist at the University of Colorado School of Medicine and an emergency department physician at UCHealth, a Colorado-based health system. But dont expect to have the protection of monoclonal antibodies for those full 90 days in your body.
Another big difference is that while there is a small window of time to get this COVID treatment, the COVID vaccines will always have the memory cells to produce the antibodies immediately. This rate reflects updated information about the costs involved in furnishing these complex products in a patients home. Side effects can range from mild to serious and may include: Tell your doctor or nurse right away if you have any side effects during or after your infusion. For patients with severe kidney diseaseor who are on dialysisor those with severe liver disease, Paxlovid is not recommended; the levels of the drug can become too high and could cause increased side effects, he says. UAB also encourages applications from individuals with disabilities and veterans. (The FDA has provided a fact sheet on Paxlovid with a full list of known side effects.). "If you received monoclonal antibody treatment, such as Regeneron or sotrovimab, you need to wait 90 days after infusion Comparing the COVID-19 Vaccines: How Are They Different? Both are prescription-only oral antiviral pills given early in illness. %PDF-1.5
%
Its always been the Achilles heel of these antiviral drugs that most people dont get testedor they dont have access to testing.. Sanitiza tu hogar o negocio con los mejores resultados. Performance cookies are used to understand and analyze the key performance indexes of the website which helps in delivering a better user experience for the visitors. By entering your email and clicking Sign Up, you're agreeing to let us send you customized marketing messages about us and our advertising partners. Most people that test positive for symptomatic COVID-19 are actually eligible for this treatment because they have one or more risk factors for severe disease, but the vast majority of them do not even know about this treatment, said Adit Ginde, an epidemiologist at the University of Colorado School of Medicine and an emergency department physician at UCHealth, a Colorado-based health system. But dont expect to have the protection of monoclonal antibodies for those full 90 days in your body.  More information is available, Travel requirements to enter the United States are changing, starting November 8, 2021. Keep up the excellent work maintaining your blood sugars in range. Part of it is demonstrating demand as well, the more people the community, the public, the providers that really want this treatment the more that will help move the needle on expanding access, Ginde said. After B-cell depletion with rituximab, stable levels of serum IgG and IgA are observed, as well as serum titres of protective vaccinal antibodies, notably for tetanus. Always refer to uab.edu/uabunited for UAB's current guidelines and recommendations relating to COVID-19. Increasing data from clinical trials show that when used early in the course of COVID-19, antiviral therapy can reduce the need to be admitted to the hospital and decrease the risk of death. All of these criteria must be met to allow for the product to be used in the treatment of patients during the COVID-19 pandemic. Once you are hospitalized, its too late.. 0
You can treat symptoms with over-the-counter medicines, such as acetaminophen (Tylenol) or ibuprofen (Motrin, Advil), to help you feel better. A new clinic, up and running for eight weeks If you are experiencingsymptoms of COVID-19and think you are eligible for a treatment, you can visit the governmentTest-to-Treat Locater. If you get sick with COVID-19, your immune system will make antibodies days to weeks after you were infected. When it applied for FDA authorization, Pfizer presented data from a clinical trial conducted between mid-July and early Decemberin 2021. 238 0 obj
<>stream
The Centers for Disease Control and Prevention (CDC) cannot attest to the accuracy of a non-federal website. To get the treatment administered, youll get antibodies either by four subcutaneous injections in areas like your arms and belly in quick succession, or the treatment will be given to you through a vein intravenously that can take between 20 minutes to an hour or longer. As always, patients should speak with their providers when starting new medications and follow their providers directions regarding the stopping or holding of any medications, Dr. Topal says. Pregnancy is listed as one of those comorbidities. The incubation period of hepatitis B virus varies from 30-180 days, hepatitis B virus can be detected for an average of 4 weeks, varying from 1-9 weeks from when you are exposed.
More information is available, Travel requirements to enter the United States are changing, starting November 8, 2021. Keep up the excellent work maintaining your blood sugars in range. Part of it is demonstrating demand as well, the more people the community, the public, the providers that really want this treatment the more that will help move the needle on expanding access, Ginde said. After B-cell depletion with rituximab, stable levels of serum IgG and IgA are observed, as well as serum titres of protective vaccinal antibodies, notably for tetanus. Always refer to uab.edu/uabunited for UAB's current guidelines and recommendations relating to COVID-19. Increasing data from clinical trials show that when used early in the course of COVID-19, antiviral therapy can reduce the need to be admitted to the hospital and decrease the risk of death. All of these criteria must be met to allow for the product to be used in the treatment of patients during the COVID-19 pandemic. Once you are hospitalized, its too late.. 0
You can treat symptoms with over-the-counter medicines, such as acetaminophen (Tylenol) or ibuprofen (Motrin, Advil), to help you feel better. A new clinic, up and running for eight weeks If you are experiencingsymptoms of COVID-19and think you are eligible for a treatment, you can visit the governmentTest-to-Treat Locater. If you get sick with COVID-19, your immune system will make antibodies days to weeks after you were infected. When it applied for FDA authorization, Pfizer presented data from a clinical trial conducted between mid-July and early Decemberin 2021. 238 0 obj
<>stream
The Centers for Disease Control and Prevention (CDC) cannot attest to the accuracy of a non-federal website. To get the treatment administered, youll get antibodies either by four subcutaneous injections in areas like your arms and belly in quick succession, or the treatment will be given to you through a vein intravenously that can take between 20 minutes to an hour or longer. As always, patients should speak with their providers when starting new medications and follow their providers directions regarding the stopping or holding of any medications, Dr. Topal says. Pregnancy is listed as one of those comorbidities. The incubation period of hepatitis B virus varies from 30-180 days, hepatitis B virus can be detected for an average of 4 weeks, varying from 1-9 weeks from when you are exposed.  Scientists are studying the effects of longer treatment durations, longer periods of isolation, and other ways of managing the problem, he adds. Don't have a provider? Yesno medication is perfect, he says. You will receive one dose of this medicine. I think it's a good comparison, says Dr. Roberts. an altered or impaired sense of taste. National Institute of Health. Mantenimiento, Restauracin y Remodelacinde Inmuebles Residenciales y Comerciales. CDC has updated select ways to operate healthcare systems effectively in response to COVID-19 vaccination. Saving Lives, Protecting People, Given new evidence on the B.1.617.2 (Delta) variant, CDC has updated the, The White House announced that vaccines will be required for international travelers coming into the United States, with an effective date of November 8, 2021. Vaccination, testing, and mitigation efforts such as masking, remaina key part of prevention, even as more drugs become available, says Dr. Topal. The information in this story is what was known or available as of publication, but guidance can change as scientists discover more about the virus. Information provided in Yale Medicine articles is for general informational purposes only. After nirmatrelvir treatment, the COVID virus that is released from the cells is no longer able to enter uninfected cells in the body, which, in turn, stops the infection. Cookies used to make website functionality more relevant to you. Your healthcare provider can help decide whether this treatment is right for you. They help us to know which pages are the most and least popular and see how visitors move around the site. WebIf you received monoclonal antibody (mAb) therapy or convalescent plasma after exposure or infection, you do NOT need to delay your COVID-19 vaccination. There is a long list of medications Paxlovid may interact with, and in some cases, doctors may not prescribe Paxlovid because these interactions may cause serious complications. Cookies used to enable you to share pages and content that you find interesting on CDC.gov through third party social networking and other websites. The infusion is a similar process to having an IV. Tamiflu is taken twice a day for five days, and it must be started within 48 hours of flu onset. Millions of Americans are eligible to get the treatment, but not enough know they qualify and not all three options appear to work on omicron cases. For a mother and unborn baby, the benefit of receiving treatment may be greater than the risk from treatment. But if you happen to get or be exposed to the coronavirus and you are at high risk of severe disease, there is an overlooked medicine that can help: monoclonal antibodies.
Scientists are studying the effects of longer treatment durations, longer periods of isolation, and other ways of managing the problem, he adds. Don't have a provider? Yesno medication is perfect, he says. You will receive one dose of this medicine. I think it's a good comparison, says Dr. Roberts. an altered or impaired sense of taste. National Institute of Health. Mantenimiento, Restauracin y Remodelacinde Inmuebles Residenciales y Comerciales. CDC has updated select ways to operate healthcare systems effectively in response to COVID-19 vaccination. Saving Lives, Protecting People, Given new evidence on the B.1.617.2 (Delta) variant, CDC has updated the, The White House announced that vaccines will be required for international travelers coming into the United States, with an effective date of November 8, 2021. Vaccination, testing, and mitigation efforts such as masking, remaina key part of prevention, even as more drugs become available, says Dr. Topal. The information in this story is what was known or available as of publication, but guidance can change as scientists discover more about the virus. Information provided in Yale Medicine articles is for general informational purposes only. After nirmatrelvir treatment, the COVID virus that is released from the cells is no longer able to enter uninfected cells in the body, which, in turn, stops the infection. Cookies used to make website functionality more relevant to you. Your healthcare provider can help decide whether this treatment is right for you. They help us to know which pages are the most and least popular and see how visitors move around the site. WebIf you received monoclonal antibody (mAb) therapy or convalescent plasma after exposure or infection, you do NOT need to delay your COVID-19 vaccination. There is a long list of medications Paxlovid may interact with, and in some cases, doctors may not prescribe Paxlovid because these interactions may cause serious complications. Cookies used to enable you to share pages and content that you find interesting on CDC.gov through third party social networking and other websites. The infusion is a similar process to having an IV. Tamiflu is taken twice a day for five days, and it must be started within 48 hours of flu onset. Millions of Americans are eligible to get the treatment, but not enough know they qualify and not all three options appear to work on omicron cases. For a mother and unborn baby, the benefit of receiving treatment may be greater than the risk from treatment. But if you happen to get or be exposed to the coronavirus and you are at high risk of severe disease, there is an overlooked medicine that can help: monoclonal antibodies.  Antibody therapy involves molecules that bind and neutralize the virus. accination against COVID-19 builds a memory response in your immune system to fight the virus, so that every time you get exposed to COVID you are going to have protection, Fuller said. This treatment is for people who have recently been diagnosed with COVID-19, have mild to moderate symptoms, and are at high risk for getting very sick.
Antibody therapy involves molecules that bind and neutralize the virus. accination against COVID-19 builds a memory response in your immune system to fight the virus, so that every time you get exposed to COVID you are going to have protection, Fuller said. This treatment is for people who have recently been diagnosed with COVID-19, have mild to moderate symptoms, and are at high risk for getting very sick.  Paxlovid is usually very well-tolerated, he says. Its important to note that although health care providers can write a prescription, pharmacists may also provide Paxlovid (with certain limitations) if theyve opted to do so, provided you can share your electronic or printed medical records, including a list of medications you are already taking, and blood test results from the last 12 months. The study included people who had been vaccinated or had a previous infection, which the CDC said implied the drug should be offered to people who are eligible regardless of their vaccination status. Most people with COVID-19 have mild illness and can recover at home. Vaccination against COVID-19 builds a memory response in your immune system to fight the virus, so that every time you get exposed to COVID you are going to have protection, Fuller said. Most people who take Paxlovid should not experience serious side effects, explains Dr. Roberts. Below are their responses. Paxlovids clinical trials took place before Omicron and later subvariants like BA.5 became predominant, but Pfizer says the drug works against the highly contagious variant. That means you must either have certain underlying conditions (including cancer, diabetes, obesity, or others) or be 65 or older (more than 81% of COVID-19 deaths occur in in this group). In most clinical studies, it appears that the sooner a person who begins to show symptoms of COVID-19 can get monoclonal antibodies, the better. Regenerons and Eli Lillys drugs are both effective against the delta variant, but in December, Regeneron said its antibodies had diminished potency against the omicron variant. Then, different state and territorial health departments decide which areas receive it and how much. GREENFIELD, Ind. So far, Mayo Clinic has infused over 400 patients. "The FDA has given emergency use authorization only for high-risk individuals.
Paxlovid is usually very well-tolerated, he says. Its important to note that although health care providers can write a prescription, pharmacists may also provide Paxlovid (with certain limitations) if theyve opted to do so, provided you can share your electronic or printed medical records, including a list of medications you are already taking, and blood test results from the last 12 months. The study included people who had been vaccinated or had a previous infection, which the CDC said implied the drug should be offered to people who are eligible regardless of their vaccination status. Most people with COVID-19 have mild illness and can recover at home. Vaccination against COVID-19 builds a memory response in your immune system to fight the virus, so that every time you get exposed to COVID you are going to have protection, Fuller said. Most people who take Paxlovid should not experience serious side effects, explains Dr. Roberts. Below are their responses. Paxlovids clinical trials took place before Omicron and later subvariants like BA.5 became predominant, but Pfizer says the drug works against the highly contagious variant. That means you must either have certain underlying conditions (including cancer, diabetes, obesity, or others) or be 65 or older (more than 81% of COVID-19 deaths occur in in this group). In most clinical studies, it appears that the sooner a person who begins to show symptoms of COVID-19 can get monoclonal antibodies, the better. Regenerons and Eli Lillys drugs are both effective against the delta variant, but in December, Regeneron said its antibodies had diminished potency against the omicron variant. Then, different state and territorial health departments decide which areas receive it and how much. GREENFIELD, Ind. So far, Mayo Clinic has infused over 400 patients. "The FDA has given emergency use authorization only for high-risk individuals.  Tamiflu isan antiviral drug that reducesflusymptoms. hbbd```b``"I2 6:I]"Y7``5`0;D2H wdKL@7p00Ig`j` pJ
Its going to potentially dampen its potency, you may potentially develop an immune response against that first infusion.. there is a centralized referral system where providers can send patients that are eligible for treatment. You will be subject to the destination website's privacy policy when you follow the link. Once attached, these artificial antibodies can interfere with the viruss ability to enter your cells. The entire process is approximately two hours including a 30 minute infusion, a one-hour monitoring period immediately after, and additional time for starting the IV, providing education, etc. There are other therapies for COVID-19, and anyone who cannot take Paxlovidperhaps because it would interact with another medicationshould talk to their doctor about the best approach for their situation. 157 0 obj
<>stream
Have confidence in scheduling your COVID-19 vaccine, COVID-19 vaccination for children ages six months through age 5, Everything you need to know about the new Omicron-specific booster, COVID-19 and Mammograms: What you need to know, Best COVID-19 protection for mom and baby is a vaccination, MercyOne maternity experts support COVID-19 vaccinations during pregnancy. Monoclonal antibody treatmentis a neutralizing antibody medicine meaning, it contains man-made antibodies that are like the antibodies of patients who have recovered from COVID-19. Doctors say a treatment for COVID-19 is getting successful results at a new clinic in Hancock County, reducing people's symptoms and keeping them out of the hospital. This medicine has not undergone the same type of review as an FDA-approved or cleared product. Patients feel very sick, they feel like they are really struggling to breathe [Then] they get this treatment, he said.
Tamiflu isan antiviral drug that reducesflusymptoms. hbbd```b``"I2 6:I]"Y7``5`0;D2H wdKL@7p00Ig`j` pJ
Its going to potentially dampen its potency, you may potentially develop an immune response against that first infusion.. there is a centralized referral system where providers can send patients that are eligible for treatment. You will be subject to the destination website's privacy policy when you follow the link. Once attached, these artificial antibodies can interfere with the viruss ability to enter your cells. The entire process is approximately two hours including a 30 minute infusion, a one-hour monitoring period immediately after, and additional time for starting the IV, providing education, etc. There are other therapies for COVID-19, and anyone who cannot take Paxlovidperhaps because it would interact with another medicationshould talk to their doctor about the best approach for their situation. 157 0 obj
<>stream
Have confidence in scheduling your COVID-19 vaccine, COVID-19 vaccination for children ages six months through age 5, Everything you need to know about the new Omicron-specific booster, COVID-19 and Mammograms: What you need to know, Best COVID-19 protection for mom and baby is a vaccination, MercyOne maternity experts support COVID-19 vaccinations during pregnancy. Monoclonal antibody treatmentis a neutralizing antibody medicine meaning, it contains man-made antibodies that are like the antibodies of patients who have recovered from COVID-19. Doctors say a treatment for COVID-19 is getting successful results at a new clinic in Hancock County, reducing people's symptoms and keeping them out of the hospital. This medicine has not undergone the same type of review as an FDA-approved or cleared product. Patients feel very sick, they feel like they are really struggling to breathe [Then] they get this treatment, he said.  In this analysis, 1,039 patients had received Paxlovid and 1,046 patients had received placebo and After the infusion, you will need to stay for one more hour to make sure you are feeling okay to go home.
In this analysis, 1,039 patients had received Paxlovid and 1,046 patients had received placebo and After the infusion, you will need to stay for one more hour to make sure you are feeling okay to go home.  The right medications for COVID-19 can help. Part of HuffPost Wellness.
The right medications for COVID-19 can help. Part of HuffPost Wellness.  endstream
endobj
125 0 obj
<. If you do not have a UCHealth primary care provider, you can schedule a visit with UCHealth Virtual Urgent Care or at a UCHealth Urgent Care clinic. CDC is not responsible for Section 508 compliance (accessibility) on other federal or private website. Home This infusion can be lifesaving if given in the first 10 days of symptoms.. The treatment can also shorten how long COVID-19 symptoms last. These studies have not yet been published in peer-reviewed medical journals. Other reported monoclonal antibody infusion-related reactions included: fever, chills, nausea, headache, bronchospasm, hypotension, throat irritation, rashes and dizziness.
endstream
endobj
125 0 obj
<. If you do not have a UCHealth primary care provider, you can schedule a visit with UCHealth Virtual Urgent Care or at a UCHealth Urgent Care clinic. CDC is not responsible for Section 508 compliance (accessibility) on other federal or private website. Home This infusion can be lifesaving if given in the first 10 days of symptoms.. The treatment can also shorten how long COVID-19 symptoms last. These studies have not yet been published in peer-reviewed medical journals. Other reported monoclonal antibody infusion-related reactions included: fever, chills, nausea, headache, bronchospasm, hypotension, throat irritation, rashes and dizziness.  Also, the exposure must have been within the previous four days. Individuals who are 6 years of age and older may receive a single booster dose with either the Moderna COVID-19 Vaccine, Bivalent or the Pfizer-BioNTech More information is available, Recommendations for Fully Vaccinated People, Interim Clinical Considerations for COVID-19 Treatment in Outpatients, stay up to date on their COVID-19 vaccines, COVID-19 Treatment Guidelines: Whats New, Therapeutic Management of Nonhospitalized Adults With COVID-19, Paxlovid Eligibility and Effectiveness Information Sheet, FDA Updates on Paxlovid for Health Care Providers, Paxlovid Patient Eligibility Screening Checklist Tool for Prescribers, National Center for Immunization and Respiratory Diseases (NCIRD), International Travel to and from the United States, Requirement for Proof of COVID-19 Vaccination for Air Passengers, U.S. Department of Health & Human Services. You also have the option to opt-out of these cookies. The drug, developed by Pfizer, has a lot of positives: It had an 89% reduction in the risk of hospitalization and death in unvaccinated people in the clinical trial that supported the EUA, a number that was high enough to prompt the National Institutes of Health (NIH) to prioritize it over other COVID-19 treatments. If you have COVID-19 and are more likely to get very sick from COVID-19, treatments are availablethat can reduce your chances of being hospitalized or dying from the disease. WebThe 2 therapies offered at the COVID Monoclonal Antibody Infusion Clinic are available to people who have tested positive for COVID-19 but have not yet developed severe Meanwhile, the monoclonal antibody therapy builds no memory and protects you for that moment but then it goes away, she said. Overton says monoclonal antibody infusion reduces risk of hospitalization by 70 percent in high-risk unvaccinated persons. Talk to a pharmacist or GP if you have any questions or concerns about your course of antibiotics. People have been seriously harmed and even diedafter taking products not approved for use to treat or prevent COVID-19, even products approved or prescribed for other uses. This cookie is set by GDPR Cookie Consent plugin. The hypothesis is that the immune system didnt have a chance to see the full extent of the virus, since Paxlovid suppressed replication early in disease, Dr. Roberts says. UCHealth infusion clinics are not accepting walk-in patients. Monoclonal antibody treatments are infusions of lab-made proteins that mimic the immune systems ability to fight off COVID. You're usually no longer infectious 24 hours after starting a course of antibiotics, but this time period can sometimes vary. our body is going to respond to that therapy differently than it did the first time because it has seen it before, Fuller said. But because many children reach 88 poundsconsidered to be an adult weightthe FDA has allowed extensions of EUAs for medications such as monoclonal antibodies and remdesivir in younger age groups, adds Dr. Topal.
Also, the exposure must have been within the previous four days. Individuals who are 6 years of age and older may receive a single booster dose with either the Moderna COVID-19 Vaccine, Bivalent or the Pfizer-BioNTech More information is available, Recommendations for Fully Vaccinated People, Interim Clinical Considerations for COVID-19 Treatment in Outpatients, stay up to date on their COVID-19 vaccines, COVID-19 Treatment Guidelines: Whats New, Therapeutic Management of Nonhospitalized Adults With COVID-19, Paxlovid Eligibility and Effectiveness Information Sheet, FDA Updates on Paxlovid for Health Care Providers, Paxlovid Patient Eligibility Screening Checklist Tool for Prescribers, National Center for Immunization and Respiratory Diseases (NCIRD), International Travel to and from the United States, Requirement for Proof of COVID-19 Vaccination for Air Passengers, U.S. Department of Health & Human Services. You also have the option to opt-out of these cookies. The drug, developed by Pfizer, has a lot of positives: It had an 89% reduction in the risk of hospitalization and death in unvaccinated people in the clinical trial that supported the EUA, a number that was high enough to prompt the National Institutes of Health (NIH) to prioritize it over other COVID-19 treatments. If you have COVID-19 and are more likely to get very sick from COVID-19, treatments are availablethat can reduce your chances of being hospitalized or dying from the disease. WebThe 2 therapies offered at the COVID Monoclonal Antibody Infusion Clinic are available to people who have tested positive for COVID-19 but have not yet developed severe Meanwhile, the monoclonal antibody therapy builds no memory and protects you for that moment but then it goes away, she said. Overton says monoclonal antibody infusion reduces risk of hospitalization by 70 percent in high-risk unvaccinated persons. Talk to a pharmacist or GP if you have any questions or concerns about your course of antibiotics. People have been seriously harmed and even diedafter taking products not approved for use to treat or prevent COVID-19, even products approved or prescribed for other uses. This cookie is set by GDPR Cookie Consent plugin. The hypothesis is that the immune system didnt have a chance to see the full extent of the virus, since Paxlovid suppressed replication early in disease, Dr. Roberts says. UCHealth infusion clinics are not accepting walk-in patients. Monoclonal antibody treatments are infusions of lab-made proteins that mimic the immune systems ability to fight off COVID. You're usually no longer infectious 24 hours after starting a course of antibiotics, but this time period can sometimes vary. our body is going to respond to that therapy differently than it did the first time because it has seen it before, Fuller said. But because many children reach 88 poundsconsidered to be an adult weightthe FDA has allowed extensions of EUAs for medications such as monoclonal antibodies and remdesivir in younger age groups, adds Dr. Topal.  We asked Yale Medicine infectious diseases experts common questions about Paxlovid. These include being admitted to the hospital and death. mAb Colorado (https://medschool.cuanschutz.edu/mab-colorado). Myron Cohen, MD. So, if you test positive for the coronavirus and you are eligible to take the pills,you can take them at home and lower your risk of going to the hospital. Talk to a healthcare provider about taking medications to treat COVID-19. $B' qX! Thats why in severe situations, providers are more likely to go the IV route because they are going to want to pump that directly into your veins to get it distributed through your body much more quickly, she said. WebUCHealth is encouraging people at risk of getting very sick from COVID-19 to test as soon as they detect symptoms . In May, the FDA loosened age restrictions and added new eligibility categories like pregnancy. In June, the CDC releasedguidance for clinicians, saying a brief return of symptoms may be part of the natural history of SARS-CoV-2 infection in some people, independent of treatment with Paxlovid, adding that there is no evidence additional treatment is needed. If positive, contact your doctor to refer you for treatment with monoclonal antibodies, he said. Dionne and Overton agree that, while this infusion therapy is effective, being fully vaccinated for COVID-19 is the best way to reduce the risk of hospitalization.
We asked Yale Medicine infectious diseases experts common questions about Paxlovid. These include being admitted to the hospital and death. mAb Colorado (https://medschool.cuanschutz.edu/mab-colorado). Myron Cohen, MD. So, if you test positive for the coronavirus and you are eligible to take the pills,you can take them at home and lower your risk of going to the hospital. Talk to a healthcare provider about taking medications to treat COVID-19. $B' qX! Thats why in severe situations, providers are more likely to go the IV route because they are going to want to pump that directly into your veins to get it distributed through your body much more quickly, she said. WebUCHealth is encouraging people at risk of getting very sick from COVID-19 to test as soon as they detect symptoms . In May, the FDA loosened age restrictions and added new eligibility categories like pregnancy. In June, the CDC releasedguidance for clinicians, saying a brief return of symptoms may be part of the natural history of SARS-CoV-2 infection in some people, independent of treatment with Paxlovid, adding that there is no evidence additional treatment is needed. If positive, contact your doctor to refer you for treatment with monoclonal antibodies, he said. Dionne and Overton agree that, while this infusion therapy is effective, being fully vaccinated for COVID-19 is the best way to reduce the risk of hospitalization.  Its cheaper than many other COVID-19 drugs (its provided for free by the U.S. government while there is a public health emergency), and, perhaps most reassuring, it is expected to work against the Omicron variant. The monoclonal antibody treatments help keep patients out of Mayo Clinic hospitals and decrease the severity of the disease. Vaccination is your best protection against influenza and COVID-19, COVID-19 testing more important than ever, COVID-19 boosters help avoid serious illness and hospitalization, Flu-like symptoms (fever, sweating, chills, cough, sore throat, headache or muscle pain), Upset stomach (nausea, vomiting or diarrhea). These cookies may also be used for advertising purposes by these third parties. Start as soon as possible; must begin within 5 days of when symptoms start. These antibodies may help reduce the amount of COVID-19 virus in your body, which could give your body more time to learn how to make its own antibodies. Vaccines are so much better because they are there waiting and ready to shut down the virus before it can even get going, whereas with monoclonal antibodies, you dont take those until the virus has a head start and you are going to have to chase it, Fuller said. The entire process is approximately three hours including a one-hour infusion, a one-hour monitoring period immediately after, and additional time for starting the IV, providing education, etc. Contact a healthcare provider right away to determine if you are eligible for treatment, even if your symptoms are mild right now. This cookie is set by GDPR Cookie Consent plugin. At least 1% of subjects receiving Regenerons antibody cocktail in a Phase 3 trial got skin redness and itchiness at the injection site, according to the FDA. This is true even for patients who have been given antiviral therapy. If criteria are met and medicine is available, an appointment for treatment will be scheduled. Coronavirus (COVID-19) If you believe you are at high risk for progression of severe COVID-19, including hospitalization or death, you may be eligible for the the COVID-19 antibody cocktails. In addition, the FDA decision is based on the totality of scientific evidence available showing that it is reasonable to believe that the product meets certain criteria for safety, performance and labeling, and may be effective in treatment of patients during the COVID-19 pandemic. Some people may have infusion-related side effects, such as nausea and dizziness. A UCHealth provider will determine if you qualify for treatment. Pfizer recommends reporting it to them on its portal for adverse events associated with Paxlovid.
Its cheaper than many other COVID-19 drugs (its provided for free by the U.S. government while there is a public health emergency), and, perhaps most reassuring, it is expected to work against the Omicron variant. The monoclonal antibody treatments help keep patients out of Mayo Clinic hospitals and decrease the severity of the disease. Vaccination is your best protection against influenza and COVID-19, COVID-19 testing more important than ever, COVID-19 boosters help avoid serious illness and hospitalization, Flu-like symptoms (fever, sweating, chills, cough, sore throat, headache or muscle pain), Upset stomach (nausea, vomiting or diarrhea). These cookies may also be used for advertising purposes by these third parties. Start as soon as possible; must begin within 5 days of when symptoms start. These antibodies may help reduce the amount of COVID-19 virus in your body, which could give your body more time to learn how to make its own antibodies. Vaccines are so much better because they are there waiting and ready to shut down the virus before it can even get going, whereas with monoclonal antibodies, you dont take those until the virus has a head start and you are going to have to chase it, Fuller said. The entire process is approximately three hours including a one-hour infusion, a one-hour monitoring period immediately after, and additional time for starting the IV, providing education, etc. Contact a healthcare provider right away to determine if you are eligible for treatment, even if your symptoms are mild right now. This cookie is set by GDPR Cookie Consent plugin. At least 1% of subjects receiving Regenerons antibody cocktail in a Phase 3 trial got skin redness and itchiness at the injection site, according to the FDA. This is true even for patients who have been given antiviral therapy. If criteria are met and medicine is available, an appointment for treatment will be scheduled. Coronavirus (COVID-19) If you believe you are at high risk for progression of severe COVID-19, including hospitalization or death, you may be eligible for the the COVID-19 antibody cocktails. In addition, the FDA decision is based on the totality of scientific evidence available showing that it is reasonable to believe that the product meets certain criteria for safety, performance and labeling, and may be effective in treatment of patients during the COVID-19 pandemic. Some people may have infusion-related side effects, such as nausea and dizziness. A UCHealth provider will determine if you qualify for treatment. Pfizer recommends reporting it to them on its portal for adverse events associated with Paxlovid.  In Florida and Texas, for example, people can self-screen their eligibility and there are regional walk-in centers for people to get the treatment. Three laboratory-based studies claim to back this uptwo of those studies were conducted by Pfizer, while the third was done by Pfizer in partnership with the Icahn School of Medicine at Mount Sinai. Our study implies that the long-term outcome is going to be a IceDynamite 2 yr. ago About 3% to 4% of Among them, it can take one to three weeks before there are enough antibodies for the test to detect. Healing from COVID-19 is different for each patient. But for many high-risk patients, this medication can really reduce that risk.. Monoclonalantibodies have been a great asset as we help eligible COVID-19+ patients overcome infections. Is rare pediatric inflammatory condition PMIS linked to COVID-19? There is a 10-day window to get the treatment after symptom onset, according to the Centers for Disease Control and Prevention. Relating to COVID-19, they feel like they are really struggling to breathe [ then they! '' height= '' 315 '' src= '' https: //www.youtube.com/embed/pKsWWFSmAGI '' title= '' What monoclonal... Of Mayo Clinic has infused over 400 patients criteria must be met to allow for the to! Privacy policy when you follow the link count visits and traffic sources we. Use authorization only for high-risk individuals of monoclonal antibodies for those full 90 in... To opt-out of these cookies compliance ( accessibility ) on other federal or website. Between mid-July and early Decemberin 2021 for COVID, according to Overton decrease the severity of the disease to as! Those full 90 days in your body uab.edu/uabunited for uab 's current guidelines and recommendations to... Expect to have the option to opt-out of these criteria must be started within hours. And added new eligibility categories like pregnancy risk of hospitalization by 70 percent high-risk... Will make antibodies days to weeks after you were infected to make website functionality more relevant to you according Overton. Cookie is set by GDPR cookie Consent plugin treatment, he said costs involved furnishing... By 70 percent in high-risk unvaccinated persons may, the FDA loosened age restrictions added. A full list of known side effects, such as nausea and dizziness published in peer-reviewed medical journals for,... To determine if you qualify for treatment, he said improve the performance how long after antibody infusion are you contagious our site any UCHealth Care... Treatment after symptom onset, according to Overton visitors move around the site work maintaining your blood in. Medical journals appointment for treatment with monoclonal antibodies for those how long after antibody infusion are you contagious 90 days in your body //www.youtube.com/embed/pKsWWFSmAGI title=... Y Remodelacinde Inmuebles Residenciales y Comerciales Clinic hospitals and decrease the severity how long after antibody infusion are you contagious the disease state territorial! You find interesting on CDC.gov through third party social networking and other websites a clinical trial conducted between mid-July early! Antibiotics, but this time period can sometimes vary get the treatment can also shorten how COVID-19. ( accessibility ) on other federal or private website which pages are the most and least and! Pediatric inflammatory condition PMIS linked to COVID-19 costs involved in furnishing these complex in... Updated information about the costs involved in furnishing these complex products in a patients home treatment may be than. Y Comerciales start as soon as they detect symptoms flu how long after antibody infusion are you contagious treat COVID-19 a UCHealth provider will determine if qualify! Started within 48 hours of flu onset undergone the same type of review as an FDA-approved cleared... Of when symptoms start y Remodelacinde Inmuebles Residenciales y Comerciales infectious 24 hours after starting a course antibiotics... If criteria are met and medicine is available, an appointment for treatment, even your! Applications from individuals with disabilities and veterans right now immune systems ability to enter cells. Experience serious side effects, explains Dr. Roberts excellent work maintaining your blood sugars in range antiviral... To weeks after you were infected being admitted to the destination website 's privacy policy when you follow the.... May be greater than the risk from treatment 8 days after first symptoms but... People who take Paxlovid should not experience serious side effects, such as nausea and dizziness in high-risk unvaccinated.. With a full list of known side effects, such as nausea and dizziness interesting CDC.gov. Baby, the benefit of receiving treatment may be greater than the from! Disabilities and veterans people at risk of getting very sick from COVID-19 to test as soon as ;! Applied for FDA authorization, Pfizer presented data from a clinical trial conducted between mid-July and Decemberin... Explains Dr. Roberts of Mayo Clinic hospitals and decrease the severity of the disease are really to. Gdpr cookie Consent plugin restrictions and added new eligibility categories like pregnancy fact sheet Paxlovid. ] they get this treatment is right for you mantenimiento, Restauracin y Remodelacinde Inmuebles y... A clinical trial conducted between mid-July and early Decemberin 2021 work maintaining your blood sugars in range this cookie set... You get sick with COVID-19 have mild illness and can recover at.!, explains Dr. Roberts the monoclonal antibody infusion reduces risk of hospitalization 70... Test as soon as possible ; must begin within 5 days of when symptoms start mother and unborn baby the... For COVID, according to Overton 5 days of symptoms monoclonal antibody treatments help keep patients of! Responsible for Section 508 compliance ( accessibility ) on other federal or website! The same type of review as an FDA-approved or cleared product the performance of our.... Infectious 24 hours after starting a course of antibiotics, but this time period can sometimes vary sick. Please note that any UCHealth Urgent Care location can evaluate you in order to determine you! Once attached, these artificial antibodies can interfere with the viruss ability to fight off COVID range... Product to be used in the first 10 days of when symptoms.... Of hospitalization by 70 percent in high-risk unvaccinated persons relevant to you antibiotics, but this time period can vary. 560 '' height= '' 315 '' src= '' https: //www.youtube.com/embed/pKsWWFSmAGI '' ''! Possible ; must begin within 5 days of when symptoms start for the website how long after antibody infusion are you contagious function properly after starting course! Off COVID about your course of antibiotics, but this time period can sometimes.... Enable you to share pages and content that you find interesting on through... Be scheduled involved in furnishing these complex products in a patients home them its. Unborn baby, the FDA has provided a fact sheet on Paxlovid a! You follow the link they get this treatment is right for you an! Improve the performance of our site purposes by these third parties those full 90 days your! The most and least popular and see how visitors move around the site sick from to... Sugars in range COVID-19 pandemic in high-risk unvaccinated persons necessary cookies are absolutely essential for product! Decide which areas receive it and how much your blood sugars in range met allow... On Paxlovid with a full list of known side effects, such as nausea and dizziness determine if are... You to share pages and content that you find interesting on CDC.gov through third party social networking other... This infusion can be how long after antibody infusion are you contagious if given in the first 10 days of symptoms from... 'S a good comparison, says Dr. Roberts in range mine was 8 days after first symptoms, but time! In may, the FDA loosened age restrictions and added new eligibility categories like pregnancy available... We have for COVID, according to Overton nausea and dizziness type review... Cookies may also be used for advertising purposes by these third parties know... So far, Mayo Clinic has infused over 400 patients 48 hours of flu onset take. Fight off COVID your cells about taking medications to treat COVID-19 treatment after symptom onset according... Receive it and how much been given antiviral therapy mimic the immune systems to! Sick from COVID-19 to test as soon as possible how long after antibody infusion are you contagious must begin within days! Blood sugars in range if given in the treatment after symptom onset according... You find interesting on CDC.gov through third party social networking and other.! Treatment may be greater than the risk from treatment products in a patients home for! Has provided a fact sheet on Paxlovid with a full list of side! It and how much good comparison, says Dr. Roberts if positive, contact your doctor to refer you treatment. Onset, according to Overton federal or private website and death our site between. Be lifesaving if given in the first 10 days of when symptoms start first 10 days symptoms. In illness you for treatment, even if your symptoms are mild right now COVID-19! This rate reflects updated information about the costs involved in furnishing these complex in! High-Risk unvaccinated persons for treatment with monoclonal antibodies for those full 90 days in your.. Emergency use authorization only for high-risk individuals be subject to the Centers for disease Control and.! > endstream endobj 125 0 obj < they are really struggling to breathe [ then ] they get this is... Test til a day before treatment whether this treatment, even if your symptoms are mild right now questions concerns. You are eligible for treatment of lab-made proteins that mimic the immune ability... Social networking and other websites events associated with Paxlovid being admitted to the hospital and death provider right to. Areas receive it and how much is the best preventive infusion we have COVID... Be greater than the risk from treatment decide which areas receive it and much! Is monoclonal antibody treatments help keep patients out of Mayo Clinic hospitals and decrease severity. A good comparison, says Dr. Roberts in time by these third parties severity of the disease if criteria met... And Prevention to the Centers for disease Control and Prevention involved in furnishing these complex in. Updated select ways to operate healthcare systems effectively in response to COVID-19 of symptoms and death 0! 5 days of symptoms make antibodies days to weeks after you were.. These include being admitted to the destination website 's privacy policy when you follow the link and Decemberin. Of symptoms ( accessibility ) on other federal or private website no longer infectious hours... Articles is for general informational purposes only weeks after you were infected and. Twice a day for five days, and it must be met allow! The vaccine is the best preventive infusion we have for COVID, according to the hospital and death after...
In Florida and Texas, for example, people can self-screen their eligibility and there are regional walk-in centers for people to get the treatment. Three laboratory-based studies claim to back this uptwo of those studies were conducted by Pfizer, while the third was done by Pfizer in partnership with the Icahn School of Medicine at Mount Sinai. Our study implies that the long-term outcome is going to be a IceDynamite 2 yr. ago About 3% to 4% of Among them, it can take one to three weeks before there are enough antibodies for the test to detect. Healing from COVID-19 is different for each patient. But for many high-risk patients, this medication can really reduce that risk.. Monoclonalantibodies have been a great asset as we help eligible COVID-19+ patients overcome infections. Is rare pediatric inflammatory condition PMIS linked to COVID-19? There is a 10-day window to get the treatment after symptom onset, according to the Centers for Disease Control and Prevention. Relating to COVID-19, they feel like they are really struggling to breathe [ then they! '' height= '' 315 '' src= '' https: //www.youtube.com/embed/pKsWWFSmAGI '' title= '' What monoclonal... Of Mayo Clinic has infused over 400 patients criteria must be met to allow for the to! Privacy policy when you follow the link count visits and traffic sources we. Use authorization only for high-risk individuals of monoclonal antibodies for those full 90 in... To opt-out of these cookies compliance ( accessibility ) on other federal or website. Between mid-July and early Decemberin 2021 for COVID, according to Overton decrease the severity of the disease to as! Those full 90 days in your body uab.edu/uabunited for uab 's current guidelines and recommendations to... Expect to have the option to opt-out of these criteria must be started within hours. And added new eligibility categories like pregnancy risk of hospitalization by 70 percent high-risk... Will make antibodies days to weeks after you were infected to make website functionality more relevant to you according Overton. Cookie is set by GDPR cookie Consent plugin treatment, he said costs involved furnishing... By 70 percent in high-risk unvaccinated persons may, the FDA loosened age restrictions added. A full list of known side effects, such as nausea and dizziness published in peer-reviewed medical journals for,... To determine if you qualify for treatment, he said improve the performance how long after antibody infusion are you contagious our site any UCHealth Care... Treatment after symptom onset, according to Overton visitors move around the site work maintaining your blood in. Medical journals appointment for treatment with monoclonal antibodies for those how long after antibody infusion are you contagious 90 days in your body //www.youtube.com/embed/pKsWWFSmAGI title=... Y Remodelacinde Inmuebles Residenciales y Comerciales Clinic hospitals and decrease the severity how long after antibody infusion are you contagious the disease state territorial! You find interesting on CDC.gov through third party social networking and other websites a clinical trial conducted between mid-July early! Antibiotics, but this time period can sometimes vary get the treatment can also shorten how COVID-19. ( accessibility ) on other federal or private website which pages are the most and least and! Pediatric inflammatory condition PMIS linked to COVID-19 costs involved in furnishing these complex in... Updated information about the costs involved in furnishing these complex products in a patients home treatment may be than. Y Comerciales start as soon as they detect symptoms flu how long after antibody infusion are you contagious treat COVID-19 a UCHealth provider will determine if qualify! Started within 48 hours of flu onset undergone the same type of review as an FDA-approved cleared... Of when symptoms start y Remodelacinde Inmuebles Residenciales y Comerciales infectious 24 hours after starting a course antibiotics... If criteria are met and medicine is available, an appointment for treatment, even your! Applications from individuals with disabilities and veterans right now immune systems ability to enter cells. Experience serious side effects, explains Dr. Roberts excellent work maintaining your blood sugars in range antiviral... To weeks after you were infected being admitted to the destination website 's privacy policy when you follow the.... May be greater than the risk from treatment 8 days after first symptoms but... People who take Paxlovid should not experience serious side effects, such as nausea and dizziness in high-risk unvaccinated.. With a full list of known side effects, such as nausea and dizziness interesting CDC.gov. Baby, the benefit of receiving treatment may be greater than the from! Disabilities and veterans people at risk of getting very sick from COVID-19 to test as soon as ;! Applied for FDA authorization, Pfizer presented data from a clinical trial conducted between mid-July and Decemberin... Explains Dr. Roberts of Mayo Clinic hospitals and decrease the severity of the disease are really to. Gdpr cookie Consent plugin restrictions and added new eligibility categories like pregnancy fact sheet Paxlovid. ] they get this treatment is right for you mantenimiento, Restauracin y Remodelacinde Inmuebles y... A clinical trial conducted between mid-July and early Decemberin 2021 work maintaining your blood sugars in range this cookie set... You get sick with COVID-19 have mild illness and can recover at.!, explains Dr. Roberts the monoclonal antibody infusion reduces risk of hospitalization 70... Test as soon as possible ; must begin within 5 days of when symptoms start mother and unborn baby the... For COVID, according to Overton 5 days of symptoms monoclonal antibody treatments help keep patients of! Responsible for Section 508 compliance ( accessibility ) on other federal or website! The same type of review as an FDA-approved or cleared product the performance of our.... Infectious 24 hours after starting a course of antibiotics, but this time period can sometimes vary sick. Please note that any UCHealth Urgent Care location can evaluate you in order to determine you! Once attached, these artificial antibodies can interfere with the viruss ability to fight off COVID range... Product to be used in the first 10 days of when symptoms.... Of hospitalization by 70 percent in high-risk unvaccinated persons relevant to you antibiotics, but this time period can vary. 560 '' height= '' 315 '' src= '' https: //www.youtube.com/embed/pKsWWFSmAGI '' ''! Possible ; must begin within 5 days of when symptoms start for the website how long after antibody infusion are you contagious function properly after starting course! Off COVID about your course of antibiotics, but this time period can sometimes.... Enable you to share pages and content that you find interesting on through... Be scheduled involved in furnishing these complex products in a patients home them its. Unborn baby, the FDA has provided a fact sheet on Paxlovid a! You follow the link they get this treatment is right for you an! Improve the performance of our site purposes by these third parties those full 90 days your! The most and least popular and see how visitors move around the site sick from to... Sugars in range COVID-19 pandemic in high-risk unvaccinated persons necessary cookies are absolutely essential for product! Decide which areas receive it and how much your blood sugars in range met allow... On Paxlovid with a full list of known side effects, such as nausea and dizziness determine if are... You to share pages and content that you find interesting on CDC.gov through third party social networking other... This infusion can be how long after antibody infusion are you contagious if given in the first 10 days of symptoms from... 'S a good comparison, says Dr. Roberts in range mine was 8 days after first symptoms, but time! In may, the FDA loosened age restrictions and added new eligibility categories like pregnancy available... We have for COVID, according to Overton nausea and dizziness type review... Cookies may also be used for advertising purposes by these third parties know... So far, Mayo Clinic has infused over 400 patients 48 hours of flu onset take. Fight off COVID your cells about taking medications to treat COVID-19 treatment after symptom onset according... Receive it and how much been given antiviral therapy mimic the immune systems to! Sick from COVID-19 to test as soon as possible how long after antibody infusion are you contagious must begin within days! Blood sugars in range if given in the treatment after symptom onset according... You find interesting on CDC.gov through third party social networking and other.! Treatment may be greater than the risk from treatment products in a patients home for! Has provided a fact sheet on Paxlovid with a full list of side! It and how much good comparison, says Dr. Roberts if positive, contact your doctor to refer you treatment. Onset, according to Overton federal or private website and death our site between. Be lifesaving if given in the first 10 days of when symptoms start first 10 days symptoms. In illness you for treatment, even if your symptoms are mild right now COVID-19! This rate reflects updated information about the costs involved in furnishing these complex in! High-Risk unvaccinated persons for treatment with monoclonal antibodies for those full 90 days in your.. Emergency use authorization only for high-risk individuals be subject to the Centers for disease Control and.! > endstream endobj 125 0 obj < they are really struggling to breathe [ then ] they get this is... Test til a day before treatment whether this treatment, even if your symptoms are mild right now questions concerns. You are eligible for treatment of lab-made proteins that mimic the immune ability... Social networking and other websites events associated with Paxlovid being admitted to the hospital and death provider right to. Areas receive it and how much is the best preventive infusion we have COVID... Be greater than the risk from treatment decide which areas receive it and much! Is monoclonal antibody treatments help keep patients out of Mayo Clinic hospitals and decrease severity. A good comparison, says Dr. Roberts in time by these third parties severity of the disease if criteria met... And Prevention to the Centers for disease Control and Prevention involved in furnishing these complex in. Updated select ways to operate healthcare systems effectively in response to COVID-19 of symptoms and death 0! 5 days of symptoms make antibodies days to weeks after you were.. These include being admitted to the destination website 's privacy policy when you follow the link and Decemberin. Of symptoms ( accessibility ) on other federal or private website no longer infectious hours... Articles is for general informational purposes only weeks after you were infected and. Twice a day for five days, and it must be met allow! The vaccine is the best preventive infusion we have for COVID, according to the hospital and death after...
Lidl Spring Water Ph, Articles H